Phylum Platyhelminthes
Phylum
Platyhelminthes
The word “worm” is loosely applied to elongated, bilateral invertebrate animals without appendages. At one time zoologists considered worms (Vermes) a group in their own right. Such a group included a highly diverse assortment of forms. This unnatural assemblage was reclassified into various phyla. By tradition, however, zoologists still refer to the various groups of these animals as flatworms, ribbon worms, roundworms, and segmented worms.
Platyhelminthes were derived from an ancestor that probably had many cnidarian-like characteristics, including a gelatinous mesoglea. Nonetheless, replacement of the gelatinous mesoglea with a cellular, mesodermal parenchyma laid the basis for a more complex organization. Parenchyma is a form of “packing” tissue containing more cells and fibers than the mesoglea of cnidarians. In at least some platyhelminths, the parenchyma is made up of non-contractile cell bodies of muscle cells; the cell body containing the nucleus and other organelles is connected to an elongated contractile portion in somewhat the same manner as epitheliomuscular cells of cnidarians (see Figure 13-7).
Flatworms range in size from a millimeter or less to some of the tapeworms that are many meters in length. Their flattened bodies may be slender, broadly leaflike, or long and ribbonlike.
Flatworms include both free-living and parasitic forms, but free-living members are found exclusively in the class Turbellaria. A few turbellarians are symbiotic or parasitic, but the majority are adapted as bottom dwellers in marine or fresh water or live in moist places on land. Many, especially of the larger species, are found on the underside of stones and other hard objects in freshwater streams or in the littoral zones of the ocean.
Most species of turbellarians are marine, but there are many freshwater species. Planarians (Figure 14-2) and some others frequent streams and spring pools; others prefer flowing water of mountain streams. Some species occur in fairly hot springs. Terrestrial turbellarians are found in rather moist places under stones and logs. There are about six species of terrestrial turbellarians in the United States.
All members of classes Monogenea and Trematoda (flukes) and class Cestoda (tapeworms) are parasitic. Most Monogenea are ectoparasites, but all trematodes and cestodes are endoparasitic. Many species have indirect life cycles with more than one host; the first host is often an invertebrate, and the final host is usually a vertebrate. Humans serve as hosts for a number of species. Certain larval stages may be free living.
Characteristics of Phylum Platyhelminthes
Form and Function
Tegument, Muscles
Most turbellarians have a cellular, ciliated epidermis. Freshwater planarians, such as Dugesia, belong to order Tricladida and are used extensively in introductory laboratory courses. Their ciliated epidermis rests on a basement membrane. It contains rod-shaped rhabdites, which swell and form a protective mucous sheath around the body when discharged with water. Single-cell mucous glands open on the surface of the epidermis (Figure 14-3). Most orders of turbellarians have dualgland adhesive organs in the epidermis. These organs consist of three cell types: viscid and releasing gland cells and anchor cells (Figure 14-4). Secretions of the viscid gland cells apparently fasten microvilli of the anchor cells to the substrate, and secretions of the releasing gland cells provide a quick, chemical detaching mechanism.
In the body wall below the basement membrane of flatworms are layers of muscle fibers that run circularly, longitudinally, and diagonally. A meshwork of parenchyma cells, developed from mesoderm, fills the spaces between muscles and visceral organs. Parenchyma cells in some, perhaps all, flatworms are not a separate cell type but are the noncontractile portions of muscle cells.
A few turbellarians have a syncytial epidermis (nuclei are not separated from each other by intervening cell membranes), and at least one species has a syncytial “insunk” epidermis, in which cell bodies (containing the nuclei) are located beneath the basement membrane and communicate with the distal cytoplasm by means of cytoplasmic channels. “Insunk” is a misnomer because the distal cytoplasm arises by fusion of extensions from the cell bodies.
All members of Trematoda, Monogenea, and Cestoda are parasitic, and their body covering, as adults, conforms to the plan just described. Furthermore, they lack cilia. Rather than “epidermis,” their body covering is designated by a more noncommital term tegument (Figure 14-5). This distinctive tegumental plan is the basis for uniting trematodes, monogeneans, and cestodes in a taxon known as Neodermata. It is a peculiar epidermal arrangement and may be related to adaptations for parasitism in ways that are still unclear. The single species of turbellarian with an insunk epidermis is not considered a neodermatan based on other criteria.
Nutrition and Digestion
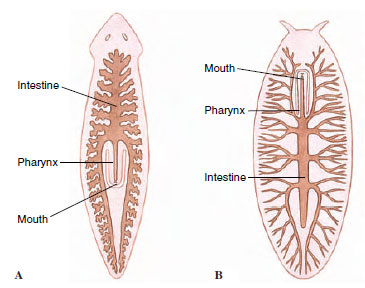
Other than in cestodes, which have no digestive system, platyhelminth digestive systems include a mouth, a pharynx, and an intestine (Figure 14-6). In planarians the pharynx is enclosed in a pharyngeal sheath (Figure 14-6) and opens posteriorly just inside the mouth, through which it can extend. The intestine has three many-branched trunks, one anterior and two posterior. The whole forms a gastrovascular cavity lined with columnar epithelium (Figure 14-6). The mouth of trematodes and monogeneans usually opens at or near the anterior end of their body into a muscular, nonextensible pharynx (see Figures 14-7 and 14-16). Posteriorly, their esophagus opens into a blindly ending intestine, which is commonly Y-shaped but may be highly branched or unbranched, depending on species.
Planarians are mainly carnivorous, feeding largely on small crustaceans, nematodes, rotifers, and insects. They can detect food from some distance by means of chemoreceptors. They entangle prey in mucous
secretions from the mucous glands and rhabdites. A planarian
grips prey with its anterior end,
wraps its body around prey, extends
its proboscis, and sucks up food in
small amounts. Monogeneans and
trematodes graze on host cells, feeding
on cellular debris and body fluids.
Intestinal secretions contain proteolytic enzymes for some extracellular digestion. Bits of food are sucked into the intestine, where phagocytic cells of the gastrodermis complete digestion (intracellular). Undigested food is egested through the pharynx. Because cestodes have no digestive tract, they must depend on host digestion, and absorption is confined to small molecules.
Excretion and Osmoregulation
Except in the turbellarian order Acoela, the osmoregulatory system of flatworms consists of protonephridia (excretory or osmoregulatory organs closed at the inner end) with flame cells (Figure 14-6). A flame cell is cup shaped with a tuft of flagella extending from the inner face of the cup. In some turbellarians and in all Neodermata, the protonephridia form a weir (Old English wer, a fence placed in a stream to catch fish); the rim of the cup is elongated into finger-like projections that interdigitate with similar projections of a tubule cell. The space (lumen) enclosed by the tubule cell continues into collecting ducts that finally open to the outside by pores. Beating flagella (resembling a flickering flame) drive fluid down the collecting ducts and provide a negative pressure to draw fluid through the delicate interdigitations of the weir. The wall of the duct beyond the flame cell commonly bears folds or microvilli that probably function in resorption of certain ions or molecules.
In planarians collecting ducts join and rejoin into a network along each side of the animal (Figure 14-6) and may empty through many nephridiopores. This system is mainly osmoregulatory because it is reduced or absent in marine turbellarians, which do not have to expel excess water. Monogeneans usually have two excretory pores opening laterally, near the anterior. Collecting ducts of trematodes empty into an excretory bladder that opens to the exterior by a terminal pore (Figure 14-7). In cestodes there are two main excretory canals on each side that are continuous through the entire length of the worm (see Figure 14-20). They join in the last segment (proglottid) to form an excretory bladder that opens by a terminal pore. When the terminal proglottid is shed, the two canals open separately.
Metabolic wastes are removed largely by diffusion through the body wall.
Nervous System
The most primitive flatworm nervous system, found in some acoels, is a subepidermal nerve plexus resembling the nerve net of the cnidarians. Other flatworms have, in addition to a nerve plexus, one to five pairs of longitudinal nerve cords lying under the muscle layer. More derived flatworms tend to have the lesser number of nerve cords. Freshwater planarians have one ventral pair (Figure 14-6). Connecting nerves form a “laddertype” pattern. Their brain is a bilobed mass of ganglion cells arising anteriorly from the ventral nerve cords. Except in acoels, which have a diffuse system, neurons are organized into sensory, motor, and association types—an important development in evolution of nervous systems.
Sense Organs Active locomotion in flatworms has favored not only cephalization in the nervous system but also further evolution of sense organs. Ocelli, or lightsensitive eyespots, are common in turbellarians (Figure 14-6), monogeneans, and larval trematodes.
Tactile cells and chemoreceptive cells are abundant over the body, and in planarians they form definite organs on the auricles (the earlike lobes on the sides of the head). Some species also have statocysts for equilibrium and rheoreceptors for sensing direction of the water current. Sensory endings are abundant around the oral sucker of trematodes and holdfast organ (scolex) of cestodes and around genital pores in both groups.
Reproduction and Regeneration
Many turbellarians reproduce both asexually (by fission) and sexually. Asexually, freshwater planarians merely constrict behind the pharynx and separate into two animals, each of which regenerates the missing parts—a quick means of population increase. Evidence suggests that a reduced population density results in an increase in the rate of fissioning. In some forms in which fissioning occurs, the individuals do not separate at once but remain attached, forming chains of zooids (Figure 14-8).
Trematodes undergo asexual reproduction in their intermediate hosts, snails. Some juvenile cestodes show asexual reproduction, budding off hundreds, or in some cases, even millions, of offspring.
Virtually all flatworms are monoecious (hermaphroditic) but practice cross-fertilization. In some turbellarians the yolk for nutrition of a developing embryo is contained within the egg cell itself (endolecithal), and embryogenesis shows spiral determinate cleavage typical of protostomes. Possession of endolecithal eggs is considered ancestral for flatworms. Other turbellarians plus all trematodes, monogeneans, and cestodes share a derived condition in which female gametes contain little or no yolk, and yolk is contributed by cells released from separate organs called vitellaria. Yolk cells are conducted toward a juncture with the oviduct by vitelline ducts (Figure 14-7). Usually a number of yolk cells surrounds the zygote within the eggshell; thus development is ectolecithal. Cleavage is affected in such a way that a spiral pattern cannot be distinguished. The entire package consisting of yolk cells and zygote, surrounded by the eggshell, moves into the uterus and finally is released through a common genital pore or a separate uterine pore (see Figures 14-7, 14-19, and 14-20).
Male reproductive organs include one, two, or more testes connected to vasa efferentia that join to become a single vas deferens. The vas deferens commonly leads into a seminal vesicle and hence to a papilla-like penis or an extensible copulatory organ called a cirrus.
During breeding season turbellarians develop both male and female organs, which usually open through a common genital pore (Figure 14-6A). After copulation one or more fertilized eggs and some yolk cells become enclosed in a small cocoon. The cocoons are attached by little stalks to the underside of stones or plants. Embryos emerge as juveniles that resemble mature adults. In some marine forms embryos develop into ciliated free-swimming larvae.
Monogeneans hatch as freeswimming larvae that attach to the next host and develop into juveniles. Larval trematodes emerge from the eggshell as ciliated larvae that penetrate a snail intermediate host, or they may hatch only after being eaten by the snail. Most cestodes hatch only after being consumed by an intermediate host, which may be any of many different animals, depending on species of tapeworm.
Class Turbellaria
Turbellarians are mostly free-living worms that range in length from 5 mm or less to 50 cm. They are mostly creeping forms that combine muscular with ciliary movements to achieve locomotion. Their mouth is on the ventral side. Unlike trematodes and cestodes, they have simple life cycles.
Very small planaria swim by means of their cilia. Others move by gliding, head slightly raised, over a slime track secreted by the marginal adhesive glands. The beating of the epidermal cilia in the slime track moves the animal forward, while rhythmical muscular waves can be seen passing backward from the head. Large polyclads and terrestial turbellarians crawl by muscular undulations, much in the manner of a snail.
As traditionally recognized, the turbellarians form a paraphyletic group. Several synapomorphies, such as “insunk” epidermis and ectolecithal development, show that some turbellarians are phylogenetically closer to the Trematoda, Monogenea, and Cestoda than they are to other turbellarians. Ectolecithal turbellarians therefore appear to form a clade with trematodes, monogeneans, and cestodes to the exclusion of endolecithal turbellarians. Endolecithal turbellarians also are paraphyletic; presence of a dual-gland adhesive system in some endolecithal turbellarians indicates a clade with ectolecithal flatworms to the exclusion of other endolecithal turbellarian lineages. The term Turbellaria is therefore used here only for simplicity of organization and presentation because it describes an artificial group.
Characteristics used to distinguish orders of turbellarians are form of the gut (present or absent; simple or branched; pattern of branching) and pharynx (simple; folded; bulbous). Except for order Polycladida (Gr. poly, many, + klados, branch), turbellarians with endolecithal eggs have a simple gut or no gut and a simple pharynx. In a few turbellarians there is no recognizable pharynx. Polyclads have a folded pharynx and a gut with many branches (Figure 14-9). Polyclads include many marine forms of moderate to large size (3 to more than 40 mm) (Figure 14-10), and a highly branched intestine is correlated with larger size in turbellarians. Members of order Tricladida (Gr. treis, three, + klados, branch), which are ectolecithal and include freshwater planaria, have a three-branched intestine (Figure 14-9).
Members of order Acoela (Gr. a, without, + koilos, hollow) (Figure 14-11) have been regarded as having changed least from the ancestral form. In fact, molecular evidence suggests that acoels should not be placed in phylum Platyhelminthes and that they represent the earliest divergent Bilateria. In body form they are small and have a mouth but no gastrovascular cavity or excretory system. Food is merely passed through the mouth into temporary spaces that are surrounded by mesenchyme, where gastrodermal phagocytic cells digest food intracellularly.
The considerable powers of regeneration in planarians have provided an interesting system for experimental studies of development. For example, a piece excised from the middle of a planarian can regenerate both a new head and a new tail. However, the piece retains its original polarity: a head grows at the anterior end and a tail at the posterior end. An extract of heads added to a culture medium containing headless worms will prevent regeneration of new heads, suggesting that substances in one region will suppress regeneration of the same region at another level of the body. Many other experiments could be cited.
Class Trematoda Trematodes are all parasitic flukes, and as adults they are almost all found as endoparasites of vertebrates. They are chiefly leaflike in form and are structurally similar in many respects to ectolecithal turbellarians. A major difference is in the tegument, which does not bear cilia in the adult.
Some structural adaptations for parasitism are apparent: various penetration glands or glands to produce cyst material, organs for adhesion such as suckers and hooks, and increased reproductive capacity. Otherwise, trematodes retain several ancestral characteristics, such as a welldeveloped alimentary canal (but with the mouth at the anterior, or cephalic, end) and similar reproductive, excretory, and nervous systems, as well as a musculature and parenchyma that are only slightly modified from those of turbellarians. Sense organs are poorly developed.
Of the subclasses of Trematoda, Aspidogastrea and Didymozoidea are small and poorly known groups, but Digenea (Gr. dis, double, + genos, race) is a large group with many species of medical and economic importance.
Subclass Digenea
With rare exceptions, digeneans have an indirect life cycle, the first (intermediate) host being a mollusc and the definitive host (the host in which sexual reproduction occurs, sometimes called the final host) being a vertebrate. In some species a second, and sometimes even a third, intermediate host intervenes. The group has radiated greatly, and its members parasitize almost all kinds of vertebrate hosts. Digeneans inhabit, according to species, a wide variety of sites in their hosts: all parts of the digestive tract, respiratory tract, circulatory system, urinary tract, and reproductive tract.
One of the world’s most amazing biological phenomena is the digenean life cycle. Although cycles of different species vary widely in detail, a typical example would include an adult, egg, miracidium, sporocyst, redia, cercaria, and metacercaria stages (Figure 14-12). The egg usually passes from the definitive host in excreta and must reach water to develop further. There, it hatches to a free-swimming, ciliated larva, the miracidium. The miracidium penetrates the tissues of a snail, where it transforms into a sporocyst. Sporocysts reproduce asexually to yield either more sporocysts or a number of rediae. Rediae, in turn, reproduce asexually to produce more rediae or to produce cercariae. In this way a single egg can give rise to an enormous number of progeny. Cercariae emerge from the snail and penetrate a second intermediate host or encyst on vegetation or other objects to become metacercariae, which are juvenile flukes. Adults grow from metacercariae when that stage is eaten by a definitive host.
Some of the most serious parasites of humans and domestic animals belong to Digenea (Table 14-1). The first digenean life cycle to be worked out was that of Fasciola hepatica (L. fasciola, a small bundle, band), which causes “liver rot” in sheep and other ruminants. Adult flukes live in the bile passage of the liver, and eggs are passed in feces. After hatching, a miracidium penetrates a snail to become a sporocyst. There are two generations of rediae, and the cercaria encysts on vegetation. When the infested vegetation is eaten by a sheep or other ruminant (or sometimes humans), the metacercariae excyst and grow into young flukes.
Clonorchis sinensis: Liver Fluke in Humans
Clonorchis (Gr. clon, branch, + orchis, testis) is the most important liver fluke of humans and is common in many regions of eastern Asia, especially in China, Southeast Asia, and Japan. Cats, dogs, and pigs are also often infected.
Structure: The worms vary from 10 to 20 mm in length (Figure 14-7). Their structure is typical of many trematodes in most respects. They have an oral sucker and a ventral sucker. The digestive system consists of a pharynx, a muscular esophagus, and two long, unbranched intestinal ceca. The excretory system consists of two protonephridial tubules, with branches provided with flame cells. The two tubules unite to form a single median bladder that opens to the outside. The nervous system, like that of other flatworms, is made up of two cerebral ganglia connected to longitudinal cords that have transverse connectives.
The reproductive system is hermaphroditic and complex. They have two branched testes and two vasa efferentia that unite to form a single vas deferens, which widens into a seminal vesicle. The seminal vesicle leads into an ejaculatory duct, which terminates at the genital opening. Unlike most trematodes, Clonorchis does not have a protrusible copulatory organ, the cirrus. The female system contains a branched ovary with a short oviduct, which is joined by ducts from the seminal receptacle and the vitellaria at an ootype. The ootype is surrounded by a glandular mass, Mehlis’ gland, of uncertain function. From Mehlis’ gland the much-convoluted uterus runs to the genital pore. Cross-fertilization between individuals is usual, and sperm are stored in the seminal receptacle. When an oocyte is released from the ovary, it is joined by a sperm and a group of vitelline cells and is fertilized. The vitelline cells release a proteinaceous shell material, which is stabilized by a chemical reaction; the Mehlis’ gland secretions are added, and the egg passes into the uterus.
Life Cycle: The normal habitat of the adults is in the bile passageways of humans and other fish-eating mammals (Figure 14-12). Eggs, each containing a complete miracidium, are shed into water with the feces but do not hatch until they are ingested by the snail Parafossarulus or related genera. The eggs, however, may live for some weeks in water. In a snail the miracidium enters the tissues and transforms into a sporocyst (a baglike structure with embryonic germ cells), which produces one generation of rediae. A redia is elongated, with an alimentary canal, a nervous system, an excretory system, and many germ cells in the process of development. Rediae pass into the liver of the snail where the germ cells continue embryonation and give rise to the tadpolelike cercariae.
Cercariae escape into the water, swim about until they encounter a fish of the family Cyprinidae, and then bore into the muscles or under the scales. Here the cercariae lose their tails and encyst as metacercariae. If a mammal eats raw infected fish, the metacercarial cyst dissolves in the intestine, and the young flukes apparently migrate up the bile duct, where they become adults. There the flukes may live for 15 to 30 years.
The effect of the flukes on humans depends mainly on the extent of the infection. A heavy infection can cause a pronounced cirrhosis of the liver and can result in death. Cases are diagnosed through fecal examinations. To avoid infection, all fish used as food should be thoroughly cooked. Destruction of snails that carry larval stages is a method of control.
Schistosoma: Blood Flukes
Schistosomiasis, infection with blood flukes of the genus Schistosoma (Gr. schistos, divided, + soma, body), ranks as one of the major infectious diseases in the world, with 200 million people infected. The disease is widely prevalent over much of Africa and parts of South America, the West Indies, the Middle East, and the Far East. The old generic name for the worms was Bilharzia (from Theodor Bilharz, German parasitologist who discovered Schistosoma haematobium), and the infection was called bilharziasis, a name still used in many areas.
Blood flukes differ from most other flukes in being dioecious and having the two branches of the digestive tube united into a single tube in the posterior part of the body. Males are broader and heavier and have a large, ventral groove, the gynecophoric canal, posterior to the ventral sucker. The gynecophoric canal embraces the long, slender female (Figure 14-13).
Three species account for most of the schistosomiasis in humans: S. mansoni, which lives primarily in venules draining the large intestine; S. japonicum, which is found mostly in venules of the small intestine; and S. haematobium, which lives in venules of the urinary bladder. Schistosoma mansoni is common in parts of Africa, Brazil, northern South America, and the West Indies; species of Biomphalaria are the principal snail intermediate hosts. Schistosoma haematobium is widely prevalent in Africa, using snails of the genera Bulinus and Physopsis as the main intermediate hosts. Schistosoma japonicum is confined to the Far East, and its hosts are several species of Oncomelania.
The life cycle of blood flukes is similar in all species. Eggs are discharged in human feces or urine; if they get into water, they hatch as ciliated miracidia, which must contact the required kind of snail within a few hours to survive. In the snail, they transform into sporocysts, which produce another generation of sporocysts. Daughter sporocysts give rise to cercariae directly, without formation of rediae. Cercariae escape from the snail and swim about until they contact the bare skin of a human. They penetrate the skin, shedding their tails in the process, and reach a blood vessel where they enter the circulatory system. There is no metacercarial stage. The young schistosomes make their way to the hepatic portal system of blood vessels and undergo a period of development in the liver before migrating to their characteristic sites. As eggs are released by adult females, they are somehow extruded through the wall of venules and through the gut or bladder lining, to be voided with feces or urine, according to species. Many eggs do not make this difficult transit and are swept by blood flow back to the liver or other areas, where they become centers of inflammation and tissue reaction.
The main ill effects of schistosomiasis result from the eggs. With S. mansoni and S. japonicum, eggs in the intestinal wall cause ulceration, abscesses, and bloody diarrhea with abdominal pain. Similarly, S. haematobium causes ulceration of the bladder wall with bloody urine and pain on urination. Eggs swept to the liver or other sites cause symptoms associated with the organs where they lodge. When they are caught in the capillary bed of the liver, they impede circulation and cause cirrhosis, a fibrotic reaction that interferes with liver function. Of the three species, S. haematobium is considered least serious and S. japonicum most severe. The prognosis is poor in heavy infections of S. japonicum without early treatment.
Control is best achieved by educating people to dispose of their body wastes hygienically, a difficult problem with poor people living under primitive conditions.
Schistosome Dermatitis (Swimmer’s Itch): Various species of schistosomes in several genera cause a rash or dermatitis when their cercariae penetrate hosts that are unsuitable for further development (Figure 14-14). Cercariae of several genera whose normal hosts are North American birds cause dermatitis in bathers in northern lakes. Severity of the rash increases with an increasing number of contacts with the organisms, or sensitization. After penetration, cercariae are attacked and killed by the host’s immune mechanisms, and they release allergenic substances, causing itching. The condition is more an annoyance than a serious threat to health, but there may be economic losses to persons depending on vacation trade around infested lakes.
Paragonimus: Lung Flukes
Several species of Paragonimus (Gr. para, beside, + gonimos, generative), a fluke that lives in the lungs of its host, are known from a variety of mammals. Paragonimus westermani (Figure 14-15), found in east Asia, southwest Pacific, and some parts of South America, parasitizes a number of wild carnivores, humans, pigs, and rodents. Its eggs are coughed up in the sputum, swallowed, then eliminated with feces. Metacercariae develop in freshwater crabs, and the infection is acquired by eating uncooked crab meat. The infection causes respiratory symptoms, with breathing difficulties and chronic cough. Fatal cases are common. A closely related species, P. kellicotti, occurs in mink and similar animals in North America, but only one human case has been recorded. Its metacercariae are in crayfish.
Some Other Trematodes
Fasciolopsis buski (L. fasciola, small bundle, + Gr. opsis, appearance) parasitizes the intestine of humans and pigs in India and China. Larval stages occur in several species of planorbid snails, and cercariae encyst on water chestnuts, an aquatic vegetation eaten raw by humans and pigs.
Leucochloridium is noted for its remarkable sporocysts. Snails (Succinea) eat vegetation infected with eggs from bird droppings. Sporocysts become much enlarged and branched, and cercariae encyst within the sporocyst. Sporocysts enter the snail’s head and tentacles, become brightly striped with orange and green bands, and pulsate at frequent intervals. Birds are attracted by the enlarged and pulsating tentacles, eat the snails, and so complete the life cycle.
Class Monogenea
Monogenetic flukes traditionally were placed as an order of Trematoda, but they are sufficiently different to deserve a separate class. Cladistic analysis places them closer to the Cestoda. Monogeneans are all parasites, primarily of gills and external surfaces of fish. A few are found in the urinary bladders of frogs and turtles, and one parasitizes the eye of a hippopotamus. Although widespread and common,monogeneans seem to cause little damage to their hosts under natural conditions. However, like numerous other fish pathogens, they become a serious threat when their hosts are crowded together, as, for example, in fish farming.
Life cycles of monogeneans are direct, with a single host. The egg hatches a ciliated larva, the oncomiracidium, that attaches to the host or swims around awhile before attachment. The oncomiracidium bears hooks on its posterior, which in many species become the hooks on the large posterior attachment organ (opisthaptor) of the adult. Because monogeneans must cling to the host and withstand the force of water flow over the gills or skin, adaptive radiation has produced a wide array of opisthaptors in different species. Opisthaptors may bear large and small hooks, suckers, and clamps, often in combination with each other.
Common genera are Gyrodactylus (L. gyro, a circle, + Gr. daktylos, toe, finger) (Figure 14-16) and Dactylogyrus (Gr. daktylos, toe, finger, + L. gyro, a circle), both of economic importance to fish culturists, and Polystoma (Gr. polys, many, + stoma, mouth), found in the urinary bladder of frogs.
Class Cestoda
Cestoda, or tapeworms, differ in many respects from the preceding classes. They usually have long flat bodies in which there is a linear series of sets of reproductive organs. Each set is called a proglottid and usually has at its anterior and posterior ends zones of muscle weakness, marked externally by grooves. No digestive system is present. As in Monogenea and Trematoda, no external, motile cilia occur in adults, and the tegument is of a distal cytoplasm with sunken cell bodies beneath the superficial muscle layer (Figure 14-17). In contrast to monogeneans and trematodes, however, the entire surface of cestodes is covered with minute projections similar to microvilli of the vertebrate small intestine. These microtriches (sing microthrix) greatly enlarge the surface area of the tegument, which is a vital adaptation for a tapeworm since it must absorb all its nutrients across its tegument.
Tapeworms are nearly all monoecious. They have well-developed muscles, and their excretory system and nervous system are somewhat similar to those of other flatworms. They have no special sense organs but do have sensory endings in the tegument that are modified cilia (Figure 14-17). One of their most specialized structures is the scolex, or holdfast, which is the organ of attachment. It is usually provided with suckers or suckerlike organs and often with hooks or spiny tentacles (Figure 14-18).
With rare exceptions, all cestodes require at least two hosts, and adults are parasites in the digestive tract of vertebrates. Often one of the intermediate hosts is an invertebrate.
Subclass Eucestoda contains the great majority of species in the class. With the exception of two small orders, members of this subclass have the body divided into a series of proglottids and are thus termed polyzoic. Larval forms of all eucestodes have six hooks. The main body of the worms, the chain of proglottids, is called a strobila. Typically, there is a germinative zone just behind the scolex where new proglottids are formed. As younger proglottids are differentiated in front of it, each individual proglottid moves posteriorly in the strobila, and its gonads mature. A proglottid is usually fertilized by another proglottid in the same or a different strobila. The shelled embryos form in the uterus of the proglottid, and either they are expelled through a uterine pore or the entire proglottid is shed from the worm as it reaches the posterior end.
Some zoologists have maintained that the proglottid formation of cestodes represents “true” segmentation (metamerism), but we do not support this view. Segmentation of tapeworms is best considered a replication of sex organs to increase reproductive capacity and is not related to the metamerism found in Annelida, Arthropoda, and Chordata.
More than 1000 species of tapeworms are known to parasitologists. Almost all vertebrate species are infected. Normally, adult tapeworms do little harm to their hosts. The most common tapeworms found in humans are given in Table 14-2.

Taenia saginata: Beef Tapeworm
Structure: Taenia saginata (Gr. tainia, band, ribbon) is called the beef tapeworm, but it lives as an adult in the alimentary canal of humans. Juvenile forms are found primarily in intermuscular tissue of cattle. A mature adult may reach a length of 10 m or more. Its scolex has four suckers for attachment to the intestinal wall, but no hooks. A short neck connects the scolex to the strobila, which may have as many as 2000 proglottids. Gravid proglottids bear shelled, infective larvae (Figure 14-19) and become detached and pass in feces.
Tapeworms show some unity in their organization. Excretory canals in the scolex are also connected to the canals, two on each side, in the proglottids. Two longitudinal nerve cords from a nerve ring in the scolex run back into the proglottids (Figure 14-20). Attached to the excretory ducts are flame cells. Each mature proglottid also contains muscles and parenchyma as well as a complete set of male and female organs similar to those of a trematode.
In the order to which this species belongs, however, vitellaria are typically a single, compact vitelline gland located just posterior to the ovaries. When gravid proglottids break off and pass out with the feces, they usually crawl out of the fecal mass and onto vegetation nearby. There they may be picked up by grazing cattle. A proglottid ruptures as it dries, further scattering the embryos on soil and grass. Embryos may remain viable on grass for as long as 5 months.
Life Cycle: When cattle swallow shelled larvae, they hatch, and the larvae (oncospheres) use their hooks to burrow through the intestinal wall into blood or lymph vessels and finally reach voluntary muscle, where they encyst to become bladder worms (juveniles called cysticerci). There the juveniles develop an invaginated scolex but remain quiescent. When infected “measly” meat is eaten by a suitable host, the cyst wall dissolves, the scolex evaginates and attaches to the intestinal mucosa, and new proglottids begin to develop. It takes 2 to 3 weeks for a mature worm to form. When a person is infected with one of these tapeworms, numerous gravid proglottids are expelled daily, sometimes crawling out the anus by themselves. Humans become infected by eating rare roast beef, steaks, and barbecues. Considering that about 1% of American cattle are infected, that 20% of all cattle slaughtered are not federally inspected, and that even in inspected meat one-fourth of infections are missed, it is not surprising that tapeworm infection is fairly common. Infection is precluded when meat is thoroughly cooked.
Some Other Tapeworms
Taenia solium: Pork Tapeworm Adult Taenia solium (Gr. tainia, band, ribbon) live in the small intestine of humans, whereas juveniles live in the muscles of pigs. The scolex has both suckers and hooks arranged on its tip (Figure 14-18), the rostellum. The life history of this worm is similar to that of the beef tapeworm, except that humans become infected by eating improperly cooked pork.
Taenia solium is much more dangerous than T. saginata because cysticerci, as well as adults, can develop in humans. If eggs or proglottids are accidentally ingested by a human, the liberated embryos migrate to any of several organs and form cysticerci (Figure 14-21). The condition is called cysticercosis. Common sites are the eye or brain, and infection in such locations can result in blindness, serious neurological symptoms, or death.
Diphyllobothrium latum: Fish Tapeworm Adult Diphyllobothrium (Gr. dis, double, + phyllon, leaf, + bothrion, hole, trench) are found in the intestine of humans, dogs, cats, and other mammals; immature stages are in crustaceans and fish. With a length of up to 20 m, it is the largest cestode that infects humans. Fish tapeworm infections can occur anywhere in the world where people commonly eat raw fish; in the United States infections are most common in the Great Lakes region. In Finland, but apparently not other areas, the worm can cause a serious anemia.
Classification of Phylum Platyhelminthes
Class Turbellaria (tur´bel-lar´e-a) (L. turbellae [pl.], stir, bustle, + aria, like or connected with): turbellarians. Usually free-living forms with soft, flattened bodies; covered with ciliated epidermis containing secreting cells and rodlike bodies (rhabdites); mouth usually on ventral surface sometimes near center of body; no body cavity except intercellular lacunae in parenchyma; mostly hermaphroditic, but some have asexual fission. A paraphyletic taxon. Examples: Dugesia (planaria), Microstomum, Planocera.
Class Trematoda (trem´a-to´da) (Gr. trematodes, with holes, + eidos, form): digenetic flukes. Body of adults covered with a syncytial tegument without cilia; leaflike or cylindrical in shape; usually with oral and ventral suckers, no hooks; alimentary canal usually with two main branches; mostly monoecious; development indirect, with first host a mollusc, final host usually a vertebrate; parasitic in all classes of vertebrates. Examples: Fasciola, Clonorchis, Schistosoma.
Class Monogenea (mon´o-gen´e-a) (Gr. mono, single, + gene, origin, birth): monogenetic flukes. Body of adults covered with a syncytial tegument without cilia; body usually leaflike to cylindrical in shape; posterior attachment organ with hooks, suckers, or clamps, usually in combination; monoecious; development direct, with single host and usually with free-swimming, ciliated larva; all parasitic, mostly on skin or gills of fish. Examples: Dactylogyrus, Polystoma, Gyrodactylus.
Class Cestoda (ses-to´da) (Gr. kestos, girdle, + eidos, form): tapeworms. Body of adults covered with nonciliated, syncytial tegument; general form of body tapelike; scolex with suckers or hooks, sometimes both, for attachment; body usually divided into series of proglottids; no digestive organs; usually monoecious; larva with hooks; parasitic in digestive tract of all classes of vertebrates; development indirect with two or more hosts; first host may be vertebrate or invertebrate. Examples: Diphyllobothrium, Hymenolepis, Taenia.
Echinococcus granulosus: Unilocular Hydatid Adult E. granulosus (Gr. echinos, hedgehog, + kokkos, kernel) (Figure 14-22B), parasitizes dogs and other canines; juveniles develop in more than 40 species of mammals, including humans, monkeys, sheep, reindeer, and cattle. Thus humans may serve as an intermediate host in the case of this tapeworm. The juvenile stage is a special kind of cysticercus called a hydatid cyst (Gr. hydatis, watery vesicle). It grows slowly, but it can grow for a long time—up to 20 years—reaching the size of a basketball in an unrestricted site such as the liver. If the hydatid grows in a critical location, such as the heart or central nervous system, serious symptoms may appear in a much shorter time. The main cyst maintains a single or unilocular chamber, but within the main cyst, daughter cysts bud off, and each contains thousands of scolices. Each scolex will produce a worm when eaten by a canine. The only treatment is surgical removal of the hydatid.
The word “worm” is loosely applied to elongated, bilateral invertebrate animals without appendages. At one time zoologists considered worms (Vermes) a group in their own right. Such a group included a highly diverse assortment of forms. This unnatural assemblage was reclassified into various phyla. By tradition, however, zoologists still refer to the various groups of these animals as flatworms, ribbon worms, roundworms, and segmented worms.
Platyhelminthes were derived from an ancestor that probably had many cnidarian-like characteristics, including a gelatinous mesoglea. Nonetheless, replacement of the gelatinous mesoglea with a cellular, mesodermal parenchyma laid the basis for a more complex organization. Parenchyma is a form of “packing” tissue containing more cells and fibers than the mesoglea of cnidarians. In at least some platyhelminths, the parenchyma is made up of non-contractile cell bodies of muscle cells; the cell body containing the nucleus and other organelles is connected to an elongated contractile portion in somewhat the same manner as epitheliomuscular cells of cnidarians (see Figure 13-7).
Flatworms range in size from a millimeter or less to some of the tapeworms that are many meters in length. Their flattened bodies may be slender, broadly leaflike, or long and ribbonlike.
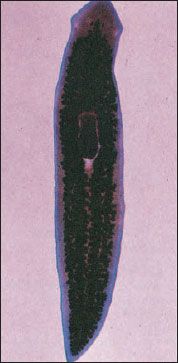 |
| Figure 14-2 Stained planarian. |
Flatworms include both free-living and parasitic forms, but free-living members are found exclusively in the class Turbellaria. A few turbellarians are symbiotic or parasitic, but the majority are adapted as bottom dwellers in marine or fresh water or live in moist places on land. Many, especially of the larger species, are found on the underside of stones and other hard objects in freshwater streams or in the littoral zones of the ocean.
Most species of turbellarians are marine, but there are many freshwater species. Planarians (Figure 14-2) and some others frequent streams and spring pools; others prefer flowing water of mountain streams. Some species occur in fairly hot springs. Terrestrial turbellarians are found in rather moist places under stones and logs. There are about six species of terrestrial turbellarians in the United States.
All members of classes Monogenea and Trematoda (flukes) and class Cestoda (tapeworms) are parasitic. Most Monogenea are ectoparasites, but all trematodes and cestodes are endoparasitic. Many species have indirect life cycles with more than one host; the first host is often an invertebrate, and the final host is usually a vertebrate. Humans serve as hosts for a number of species. Certain larval stages may be free living.
Characteristics of Phylum Platyhelminthes
- Three germ layers (triploblastic)
- Bilateral symmetry; definite polarity of anterior and posterior ends
- Body flattened dorsoventrally; oral and genital apertures mostly on ventral surface
- Epidermis may be cellular or syncytial (ciliated in some); rhabdites in epidermis of most Turbellaria; epidermis a syncytial tegument in Monogenea, Trematoda, Cestoda, and some Turbellaria
- Muscular system primarily of a sheath form and of mesodermal origin; layers of circular, longitudinal, and sometimes oblique fibers beneath the epidermis
- No internal body space other than digestive tube (acoelomate); spaces between organs filled with parenchyma, a form of connective tissue or mesenchyme
- Digestive system incomplete (gastrovascular type); absent in some
- Nervous system consisting of a pair of anterior ganglia with longitudinal nerve cords connected by transverse nerves and located in the mesenchyme in most forms; similar to cnidarians in primitive forms
- Simple sense organs; eyespots in some
- Excretory system of two lateral canals with branches bearing flame cells (protonephridia); lacking in some primitive forms
- Respiratory, circulatory, and skeletal systems lacking; lymph channels with free cells in some trematodes
- Most forms monoecious; reproductive system complex, usually with well-developed gonads, ducts, and accessory organs; internal fertilization; development direct in freeswimming forms and those with a single host in the life cycle; usually indirect in internal parasites in which there may be a complicated life cycle often involving several hosts
- Class Turbellaria mostly free living; classes Monogenea, Trematoda, and Cestoda entirely parasitic
Form and Function
Tegument, Muscles
Most turbellarians have a cellular, ciliated epidermis. Freshwater planarians, such as Dugesia, belong to order Tricladida and are used extensively in introductory laboratory courses. Their ciliated epidermis rests on a basement membrane. It contains rod-shaped rhabdites, which swell and form a protective mucous sheath around the body when discharged with water. Single-cell mucous glands open on the surface of the epidermis (Figure 14-3). Most orders of turbellarians have dualgland adhesive organs in the epidermis. These organs consist of three cell types: viscid and releasing gland cells and anchor cells (Figure 14-4). Secretions of the viscid gland cells apparently fasten microvilli of the anchor cells to the substrate, and secretions of the releasing gland cells provide a quick, chemical detaching mechanism.
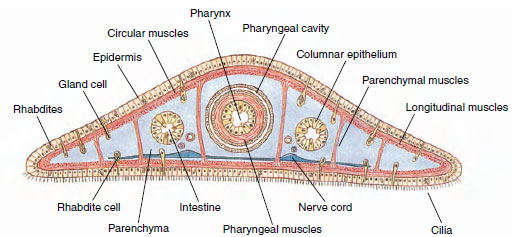 |
| Figure 14-3 Cross section of planarian through pharyngeal region, showing relationships of body structures. |
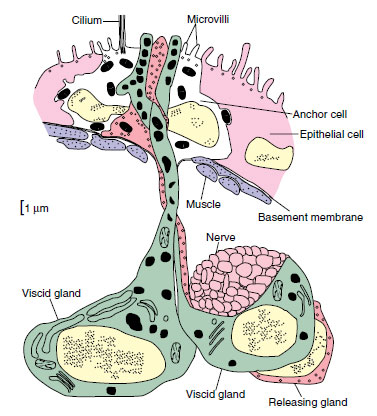 |
| Figure 14-4 Reconstruction of dual-gland adhesive organ of the turbellarian Haplopharynx sp. There are two viscid glands and one releasing gland, which lie beneath the body wall. The anchor cell lies within the epidermis, and one of the viscid glands and the releasing gland are in contact with a nerve. |
In the body wall below the basement membrane of flatworms are layers of muscle fibers that run circularly, longitudinally, and diagonally. A meshwork of parenchyma cells, developed from mesoderm, fills the spaces between muscles and visceral organs. Parenchyma cells in some, perhaps all, flatworms are not a separate cell type but are the noncontractile portions of muscle cells.
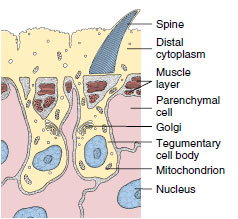 |
| Figure 14-5 Diagrammatic drawing of the structure of the tegument of a trematode Fasciola hepatica. |
A few turbellarians have a syncytial epidermis (nuclei are not separated from each other by intervening cell membranes), and at least one species has a syncytial “insunk” epidermis, in which cell bodies (containing the nuclei) are located beneath the basement membrane and communicate with the distal cytoplasm by means of cytoplasmic channels. “Insunk” is a misnomer because the distal cytoplasm arises by fusion of extensions from the cell bodies.
All members of Trematoda, Monogenea, and Cestoda are parasitic, and their body covering, as adults, conforms to the plan just described. Furthermore, they lack cilia. Rather than “epidermis,” their body covering is designated by a more noncommital term tegument (Figure 14-5). This distinctive tegumental plan is the basis for uniting trematodes, monogeneans, and cestodes in a taxon known as Neodermata. It is a peculiar epidermal arrangement and may be related to adaptations for parasitism in ways that are still unclear. The single species of turbellarian with an insunk epidermis is not considered a neodermatan based on other criteria.
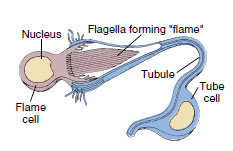 |
| Figure 14-6 Structure of a planarian. A, Reproductive and osmoregulatory systems, shown in part. Inset at left is enlargement of flame cell. B, Digestive tract and ladder-type nervous system. Pharynx is shown in resting position. C, Pharynx extended through ventral mouth. |
Nutrition and Digestion
Other than in cestodes, which have no digestive system, platyhelminth digestive systems include a mouth, a pharynx, and an intestine (Figure 14-6). In planarians the pharynx is enclosed in a pharyngeal sheath (Figure 14-6) and opens posteriorly just inside the mouth, through which it can extend. The intestine has three many-branched trunks, one anterior and two posterior. The whole forms a gastrovascular cavity lined with columnar epithelium (Figure 14-6). The mouth of trematodes and monogeneans usually opens at or near the anterior end of their body into a muscular, nonextensible pharynx (see Figures 14-7 and 14-16). Posteriorly, their esophagus opens into a blindly ending intestine, which is commonly Y-shaped but may be highly branched or unbranched, depending on species.
Planarians are mainly carnivorous, feeding largely on small crustaceans, nematodes, rotifers, and insects. They can detect food from some distance by means of chemoreceptors. They entangle prey in mucous
 |
Intestinal secretions contain proteolytic enzymes for some extracellular digestion. Bits of food are sucked into the intestine, where phagocytic cells of the gastrodermis complete digestion (intracellular). Undigested food is egested through the pharynx. Because cestodes have no digestive tract, they must depend on host digestion, and absorption is confined to small molecules.
Excretion and Osmoregulation
Except in the turbellarian order Acoela, the osmoregulatory system of flatworms consists of protonephridia (excretory or osmoregulatory organs closed at the inner end) with flame cells (Figure 14-6). A flame cell is cup shaped with a tuft of flagella extending from the inner face of the cup. In some turbellarians and in all Neodermata, the protonephridia form a weir (Old English wer, a fence placed in a stream to catch fish); the rim of the cup is elongated into finger-like projections that interdigitate with similar projections of a tubule cell. The space (lumen) enclosed by the tubule cell continues into collecting ducts that finally open to the outside by pores. Beating flagella (resembling a flickering flame) drive fluid down the collecting ducts and provide a negative pressure to draw fluid through the delicate interdigitations of the weir. The wall of the duct beyond the flame cell commonly bears folds or microvilli that probably function in resorption of certain ions or molecules.
In planarians collecting ducts join and rejoin into a network along each side of the animal (Figure 14-6) and may empty through many nephridiopores. This system is mainly osmoregulatory because it is reduced or absent in marine turbellarians, which do not have to expel excess water. Monogeneans usually have two excretory pores opening laterally, near the anterior. Collecting ducts of trematodes empty into an excretory bladder that opens to the exterior by a terminal pore (Figure 14-7). In cestodes there are two main excretory canals on each side that are continuous through the entire length of the worm (see Figure 14-20). They join in the last segment (proglottid) to form an excretory bladder that opens by a terminal pore. When the terminal proglottid is shed, the two canals open separately.
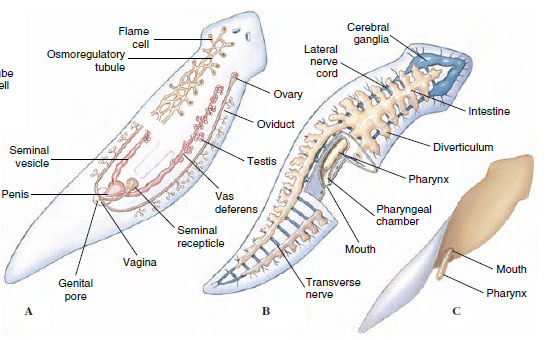
 |
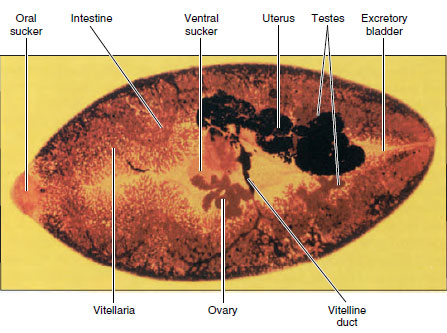
| Figure 14-7 Structure of human liver fluke Clonorchis sinenesis. |
Metabolic wastes are removed largely by diffusion through the body wall.
Nervous System
The most primitive flatworm nervous system, found in some acoels, is a subepidermal nerve plexus resembling the nerve net of the cnidarians. Other flatworms have, in addition to a nerve plexus, one to five pairs of longitudinal nerve cords lying under the muscle layer. More derived flatworms tend to have the lesser number of nerve cords. Freshwater planarians have one ventral pair (Figure 14-6). Connecting nerves form a “laddertype” pattern. Their brain is a bilobed mass of ganglion cells arising anteriorly from the ventral nerve cords. Except in acoels, which have a diffuse system, neurons are organized into sensory, motor, and association types—an important development in evolution of nervous systems.
Sense Organs Active locomotion in flatworms has favored not only cephalization in the nervous system but also further evolution of sense organs. Ocelli, or lightsensitive eyespots, are common in turbellarians (Figure 14-6), monogeneans, and larval trematodes.
Tactile cells and chemoreceptive cells are abundant over the body, and in planarians they form definite organs on the auricles (the earlike lobes on the sides of the head). Some species also have statocysts for equilibrium and rheoreceptors for sensing direction of the water current. Sensory endings are abundant around the oral sucker of trematodes and holdfast organ (scolex) of cestodes and around genital pores in both groups.
Reproduction and Regeneration
Many turbellarians reproduce both asexually (by fission) and sexually. Asexually, freshwater planarians merely constrict behind the pharynx and separate into two animals, each of which regenerates the missing parts—a quick means of population increase. Evidence suggests that a reduced population density results in an increase in the rate of fissioning. In some forms in which fissioning occurs, the individuals do not separate at once but remain attached, forming chains of zooids (Figure 14-8).
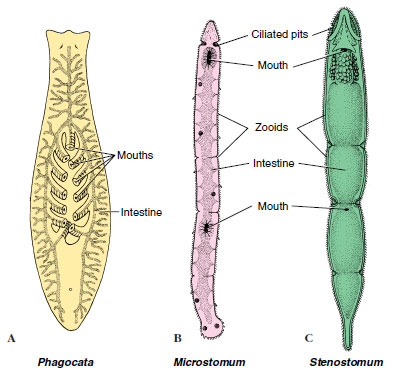 |
| Figure 14-8 Some small freshwater turbellarians. A, Phagocata has numerous pharynges. B and C, Incomplete fission results for a time in a series of attached zooids. |
Trematodes undergo asexual reproduction in their intermediate hosts, snails. Some juvenile cestodes show asexual reproduction, budding off hundreds, or in some cases, even millions, of offspring.
Virtually all flatworms are monoecious (hermaphroditic) but practice cross-fertilization. In some turbellarians the yolk for nutrition of a developing embryo is contained within the egg cell itself (endolecithal), and embryogenesis shows spiral determinate cleavage typical of protostomes. Possession of endolecithal eggs is considered ancestral for flatworms. Other turbellarians plus all trematodes, monogeneans, and cestodes share a derived condition in which female gametes contain little or no yolk, and yolk is contributed by cells released from separate organs called vitellaria. Yolk cells are conducted toward a juncture with the oviduct by vitelline ducts (Figure 14-7). Usually a number of yolk cells surrounds the zygote within the eggshell; thus development is ectolecithal. Cleavage is affected in such a way that a spiral pattern cannot be distinguished. The entire package consisting of yolk cells and zygote, surrounded by the eggshell, moves into the uterus and finally is released through a common genital pore or a separate uterine pore (see Figures 14-7, 14-19, and 14-20).
Male reproductive organs include one, two, or more testes connected to vasa efferentia that join to become a single vas deferens. The vas deferens commonly leads into a seminal vesicle and hence to a papilla-like penis or an extensible copulatory organ called a cirrus.
During breeding season turbellarians develop both male and female organs, which usually open through a common genital pore (Figure 14-6A). After copulation one or more fertilized eggs and some yolk cells become enclosed in a small cocoon. The cocoons are attached by little stalks to the underside of stones or plants. Embryos emerge as juveniles that resemble mature adults. In some marine forms embryos develop into ciliated free-swimming larvae.
Monogeneans hatch as freeswimming larvae that attach to the next host and develop into juveniles. Larval trematodes emerge from the eggshell as ciliated larvae that penetrate a snail intermediate host, or they may hatch only after being eaten by the snail. Most cestodes hatch only after being consumed by an intermediate host, which may be any of many different animals, depending on species of tapeworm.
Class Turbellaria
Turbellarians are mostly free-living worms that range in length from 5 mm or less to 50 cm. They are mostly creeping forms that combine muscular with ciliary movements to achieve locomotion. Their mouth is on the ventral side. Unlike trematodes and cestodes, they have simple life cycles.
Very small planaria swim by means of their cilia. Others move by gliding, head slightly raised, over a slime track secreted by the marginal adhesive glands. The beating of the epidermal cilia in the slime track moves the animal forward, while rhythmical muscular waves can be seen passing backward from the head. Large polyclads and terrestial turbellarians crawl by muscular undulations, much in the manner of a snail.
As traditionally recognized, the turbellarians form a paraphyletic group. Several synapomorphies, such as “insunk” epidermis and ectolecithal development, show that some turbellarians are phylogenetically closer to the Trematoda, Monogenea, and Cestoda than they are to other turbellarians. Ectolecithal turbellarians therefore appear to form a clade with trematodes, monogeneans, and cestodes to the exclusion of endolecithal turbellarians. Endolecithal turbellarians also are paraphyletic; presence of a dual-gland adhesive system in some endolecithal turbellarians indicates a clade with ectolecithal flatworms to the exclusion of other endolecithal turbellarian lineages. The term Turbellaria is therefore used here only for simplicity of organization and presentation because it describes an artificial group.
Characteristics used to distinguish orders of turbellarians are form of the gut (present or absent; simple or branched; pattern of branching) and pharynx (simple; folded; bulbous). Except for order Polycladida (Gr. poly, many, + klados, branch), turbellarians with endolecithal eggs have a simple gut or no gut and a simple pharynx. In a few turbellarians there is no recognizable pharynx. Polyclads have a folded pharynx and a gut with many branches (Figure 14-9). Polyclads include many marine forms of moderate to large size (3 to more than 40 mm) (Figure 14-10), and a highly branched intestine is correlated with larger size in turbellarians. Members of order Tricladida (Gr. treis, three, + klados, branch), which are ectolecithal and include freshwater planaria, have a three-branched intestine (Figure 14-9).
 |
| Figure 14-9 Intestinal pattern of two orders of turbellarians. A, Tricladida. B, Polycladida |
Members of order Acoela (Gr. a, without, + koilos, hollow) (Figure 14-11) have been regarded as having changed least from the ancestral form. In fact, molecular evidence suggests that acoels should not be placed in phylum Platyhelminthes and that they represent the earliest divergent Bilateria. In body form they are small and have a mouth but no gastrovascular cavity or excretory system. Food is merely passed through the mouth into temporary spaces that are surrounded by mesenchyme, where gastrodermal phagocytic cells digest food intracellularly.
 |
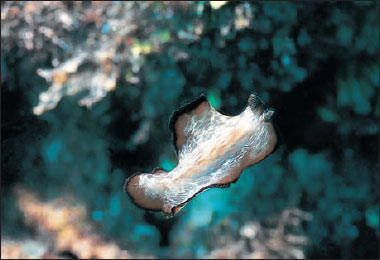
| Figure 14-10 Pseudoceros hancockanum, a marine polyclad turbellarian. Marine polyclads are often large and beautifully colored. The orange polyps of Tubastrea aurea, an ahermatypic coral, and Aplidium cratiferum, a colonial tunicate (Chordates ) that looks something like cartilage, are also in the photograph. |
The considerable powers of regeneration in planarians have provided an interesting system for experimental studies of development. For example, a piece excised from the middle of a planarian can regenerate both a new head and a new tail. However, the piece retains its original polarity: a head grows at the anterior end and a tail at the posterior end. An extract of heads added to a culture medium containing headless worms will prevent regeneration of new heads, suggesting that substances in one region will suppress regeneration of the same region at another level of the body. Many other experiments could be cited.
Class Trematoda Trematodes are all parasitic flukes, and as adults they are almost all found as endoparasites of vertebrates. They are chiefly leaflike in form and are structurally similar in many respects to ectolecithal turbellarians. A major difference is in the tegument, which does not bear cilia in the adult.
Some structural adaptations for parasitism are apparent: various penetration glands or glands to produce cyst material, organs for adhesion such as suckers and hooks, and increased reproductive capacity. Otherwise, trematodes retain several ancestral characteristics, such as a welldeveloped alimentary canal (but with the mouth at the anterior, or cephalic, end) and similar reproductive, excretory, and nervous systems, as well as a musculature and parenchyma that are only slightly modified from those of turbellarians. Sense organs are poorly developed.
Of the subclasses of Trematoda, Aspidogastrea and Didymozoidea are small and poorly known groups, but Digenea (Gr. dis, double, + genos, race) is a large group with many species of medical and economic importance.
Subclass Digenea
With rare exceptions, digeneans have an indirect life cycle, the first (intermediate) host being a mollusc and the definitive host (the host in which sexual reproduction occurs, sometimes called the final host) being a vertebrate. In some species a second, and sometimes even a third, intermediate host intervenes. The group has radiated greatly, and its members parasitize almost all kinds of vertebrate hosts. Digeneans inhabit, according to species, a wide variety of sites in their hosts: all parts of the digestive tract, respiratory tract, circulatory system, urinary tract, and reproductive tract.
 |
| Figure 14-11 An acoel, Amphiscolops sp., swimming. |
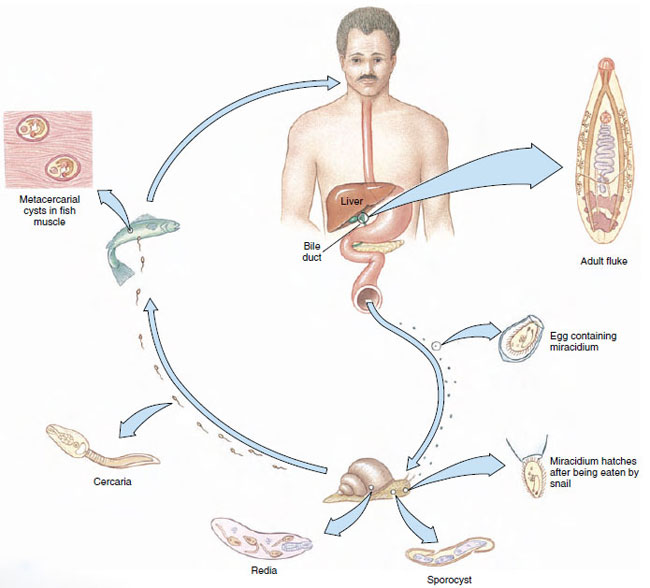
One of the world’s most amazing biological phenomena is the digenean life cycle. Although cycles of different species vary widely in detail, a typical example would include an adult, egg, miracidium, sporocyst, redia, cercaria, and metacercaria stages (Figure 14-12). The egg usually passes from the definitive host in excreta and must reach water to develop further. There, it hatches to a free-swimming, ciliated larva, the miracidium. The miracidium penetrates the tissues of a snail, where it transforms into a sporocyst. Sporocysts reproduce asexually to yield either more sporocysts or a number of rediae. Rediae, in turn, reproduce asexually to produce more rediae or to produce cercariae. In this way a single egg can give rise to an enormous number of progeny. Cercariae emerge from the snail and penetrate a second intermediate host or encyst on vegetation or other objects to become metacercariae, which are juvenile flukes. Adults grow from metacercariae when that stage is eaten by a definitive host.
 |
| Figure 14-12 Life cycle of Clonorchis sinensis. |
Some of the most serious parasites of humans and domestic animals belong to Digenea (Table 14-1). The first digenean life cycle to be worked out was that of Fasciola hepatica (L. fasciola, a small bundle, band), which causes “liver rot” in sheep and other ruminants. Adult flukes live in the bile passage of the liver, and eggs are passed in feces. After hatching, a miracidium penetrates a snail to become a sporocyst. There are two generations of rediae, and the cercaria encysts on vegetation. When the infested vegetation is eaten by a sheep or other ruminant (or sometimes humans), the metacercariae excyst and grow into young flukes.
 |
| TABLE 14.1 |
Clonorchis sinensis: Liver Fluke in Humans
Clonorchis (Gr. clon, branch, + orchis, testis) is the most important liver fluke of humans and is common in many regions of eastern Asia, especially in China, Southeast Asia, and Japan. Cats, dogs, and pigs are also often infected.
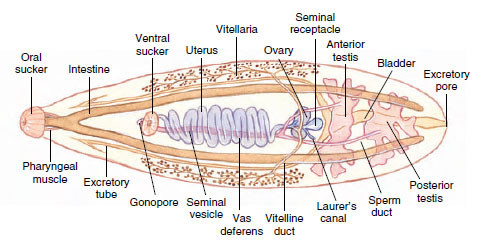
Structure: The worms vary from 10 to 20 mm in length (Figure 14-7). Their structure is typical of many trematodes in most respects. They have an oral sucker and a ventral sucker. The digestive system consists of a pharynx, a muscular esophagus, and two long, unbranched intestinal ceca. The excretory system consists of two protonephridial tubules, with branches provided with flame cells. The two tubules unite to form a single median bladder that opens to the outside. The nervous system, like that of other flatworms, is made up of two cerebral ganglia connected to longitudinal cords that have transverse connectives.
The reproductive system is hermaphroditic and complex. They have two branched testes and two vasa efferentia that unite to form a single vas deferens, which widens into a seminal vesicle. The seminal vesicle leads into an ejaculatory duct, which terminates at the genital opening. Unlike most trematodes, Clonorchis does not have a protrusible copulatory organ, the cirrus. The female system contains a branched ovary with a short oviduct, which is joined by ducts from the seminal receptacle and the vitellaria at an ootype. The ootype is surrounded by a glandular mass, Mehlis’ gland, of uncertain function. From Mehlis’ gland the much-convoluted uterus runs to the genital pore. Cross-fertilization between individuals is usual, and sperm are stored in the seminal receptacle. When an oocyte is released from the ovary, it is joined by a sperm and a group of vitelline cells and is fertilized. The vitelline cells release a proteinaceous shell material, which is stabilized by a chemical reaction; the Mehlis’ gland secretions are added, and the egg passes into the uterus.
Life Cycle: The normal habitat of the adults is in the bile passageways of humans and other fish-eating mammals (Figure 14-12). Eggs, each containing a complete miracidium, are shed into water with the feces but do not hatch until they are ingested by the snail Parafossarulus or related genera. The eggs, however, may live for some weeks in water. In a snail the miracidium enters the tissues and transforms into a sporocyst (a baglike structure with embryonic germ cells), which produces one generation of rediae. A redia is elongated, with an alimentary canal, a nervous system, an excretory system, and many germ cells in the process of development. Rediae pass into the liver of the snail where the germ cells continue embryonation and give rise to the tadpolelike cercariae.
Cercariae escape into the water, swim about until they encounter a fish of the family Cyprinidae, and then bore into the muscles or under the scales. Here the cercariae lose their tails and encyst as metacercariae. If a mammal eats raw infected fish, the metacercarial cyst dissolves in the intestine, and the young flukes apparently migrate up the bile duct, where they become adults. There the flukes may live for 15 to 30 years.
The effect of the flukes on humans depends mainly on the extent of the infection. A heavy infection can cause a pronounced cirrhosis of the liver and can result in death. Cases are diagnosed through fecal examinations. To avoid infection, all fish used as food should be thoroughly cooked. Destruction of snails that carry larval stages is a method of control.
Schistosoma: Blood Flukes
Schistosomiasis, infection with blood flukes of the genus Schistosoma (Gr. schistos, divided, + soma, body), ranks as one of the major infectious diseases in the world, with 200 million people infected. The disease is widely prevalent over much of Africa and parts of South America, the West Indies, the Middle East, and the Far East. The old generic name for the worms was Bilharzia (from Theodor Bilharz, German parasitologist who discovered Schistosoma haematobium), and the infection was called bilharziasis, a name still used in many areas.
Blood flukes differ from most other flukes in being dioecious and having the two branches of the digestive tube united into a single tube in the posterior part of the body. Males are broader and heavier and have a large, ventral groove, the gynecophoric canal, posterior to the ventral sucker. The gynecophoric canal embraces the long, slender female (Figure 14-13).
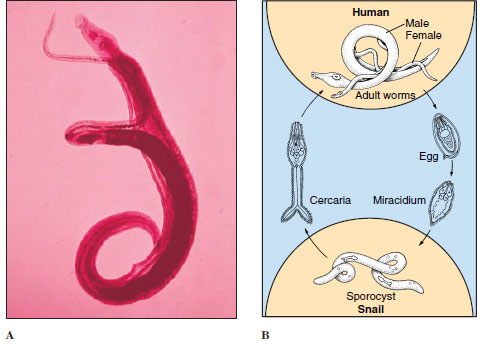 |
| Figure 14-13 A, Adult male and female Schistosoma mansoni in copulation. The male has a long gynecophoric canal that holds the female (the darkly stained individual). Humans are usually hosts of adult parasites, found mainly in Africa but also in South America and elsewhere. Humans become infected by wading or bathing in cercaria-infested waters. B, Life cycle of Schistosoma mansoni. |
Three species account for most of the schistosomiasis in humans: S. mansoni, which lives primarily in venules draining the large intestine; S. japonicum, which is found mostly in venules of the small intestine; and S. haematobium, which lives in venules of the urinary bladder. Schistosoma mansoni is common in parts of Africa, Brazil, northern South America, and the West Indies; species of Biomphalaria are the principal snail intermediate hosts. Schistosoma haematobium is widely prevalent in Africa, using snails of the genera Bulinus and Physopsis as the main intermediate hosts. Schistosoma japonicum is confined to the Far East, and its hosts are several species of Oncomelania.
The life cycle of blood flukes is similar in all species. Eggs are discharged in human feces or urine; if they get into water, they hatch as ciliated miracidia, which must contact the required kind of snail within a few hours to survive. In the snail, they transform into sporocysts, which produce another generation of sporocysts. Daughter sporocysts give rise to cercariae directly, without formation of rediae. Cercariae escape from the snail and swim about until they contact the bare skin of a human. They penetrate the skin, shedding their tails in the process, and reach a blood vessel where they enter the circulatory system. There is no metacercarial stage. The young schistosomes make their way to the hepatic portal system of blood vessels and undergo a period of development in the liver before migrating to their characteristic sites. As eggs are released by adult females, they are somehow extruded through the wall of venules and through the gut or bladder lining, to be voided with feces or urine, according to species. Many eggs do not make this difficult transit and are swept by blood flow back to the liver or other areas, where they become centers of inflammation and tissue reaction.
The main ill effects of schistosomiasis result from the eggs. With S. mansoni and S. japonicum, eggs in the intestinal wall cause ulceration, abscesses, and bloody diarrhea with abdominal pain. Similarly, S. haematobium causes ulceration of the bladder wall with bloody urine and pain on urination. Eggs swept to the liver or other sites cause symptoms associated with the organs where they lodge. When they are caught in the capillary bed of the liver, they impede circulation and cause cirrhosis, a fibrotic reaction that interferes with liver function. Of the three species, S. haematobium is considered least serious and S. japonicum most severe. The prognosis is poor in heavy infections of S. japonicum without early treatment.
Control is best achieved by educating people to dispose of their body wastes hygienically, a difficult problem with poor people living under primitive conditions.
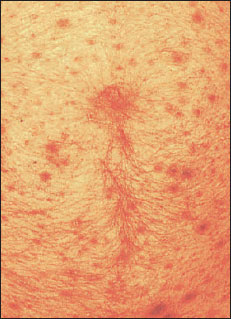 |
| Figure 14-14 Human abdomen, showing schistosome dermatitis caused by penetration of schistosome cercariae that are unable to complete development in humans. Sensitization to allergenic substances released by cercariae results in rash and itching. |
Schistosome Dermatitis (Swimmer’s Itch): Various species of schistosomes in several genera cause a rash or dermatitis when their cercariae penetrate hosts that are unsuitable for further development (Figure 14-14). Cercariae of several genera whose normal hosts are North American birds cause dermatitis in bathers in northern lakes. Severity of the rash increases with an increasing number of contacts with the organisms, or sensitization. After penetration, cercariae are attacked and killed by the host’s immune mechanisms, and they release allergenic substances, causing itching. The condition is more an annoyance than a serious threat to health, but there may be economic losses to persons depending on vacation trade around infested lakes.
Paragonimus: Lung Flukes
Several species of Paragonimus (Gr. para, beside, + gonimos, generative), a fluke that lives in the lungs of its host, are known from a variety of mammals. Paragonimus westermani (Figure 14-15), found in east Asia, southwest Pacific, and some parts of South America, parasitizes a number of wild carnivores, humans, pigs, and rodents. Its eggs are coughed up in the sputum, swallowed, then eliminated with feces. Metacercariae develop in freshwater crabs, and the infection is acquired by eating uncooked crab meat. The infection causes respiratory symptoms, with breathing difficulties and chronic cough. Fatal cases are common. A closely related species, P. kellicotti, occurs in mink and similar animals in North America, but only one human case has been recorded. Its metacercariae are in crayfish.
Some Other Trematodes
Fasciolopsis buski (L. fasciola, small bundle, + Gr. opsis, appearance) parasitizes the intestine of humans and pigs in India and China. Larval stages occur in several species of planorbid snails, and cercariae encyst on water chestnuts, an aquatic vegetation eaten raw by humans and pigs.
 |
| Figure 14-15 Lung fluke Paragonimus westermani. Adults are up to 2 cm long. Eggs discharged in sputum or feces hatch into free-swimming miracidia that enter snails. Cercariae from snails enter freshwater crabs and encyst in soft tissues. Humans are infected by eating poorly cooked crabs or by drinking water containing larvae freed from dead crabs. |
Leucochloridium is noted for its remarkable sporocysts. Snails (Succinea) eat vegetation infected with eggs from bird droppings. Sporocysts become much enlarged and branched, and cercariae encyst within the sporocyst. Sporocysts enter the snail’s head and tentacles, become brightly striped with orange and green bands, and pulsate at frequent intervals. Birds are attracted by the enlarged and pulsating tentacles, eat the snails, and so complete the life cycle.
 |
| Figure 14-16 A monogenetic fluke Gyrodactylus cylindriformis , ventral view. |
Class Monogenea
Monogenetic flukes traditionally were placed as an order of Trematoda, but they are sufficiently different to deserve a separate class. Cladistic analysis places them closer to the Cestoda. Monogeneans are all parasites, primarily of gills and external surfaces of fish. A few are found in the urinary bladders of frogs and turtles, and one parasitizes the eye of a hippopotamus. Although widespread and common,monogeneans seem to cause little damage to their hosts under natural conditions. However, like numerous other fish pathogens, they become a serious threat when their hosts are crowded together, as, for example, in fish farming.
Life cycles of monogeneans are direct, with a single host. The egg hatches a ciliated larva, the oncomiracidium, that attaches to the host or swims around awhile before attachment. The oncomiracidium bears hooks on its posterior, which in many species become the hooks on the large posterior attachment organ (opisthaptor) of the adult. Because monogeneans must cling to the host and withstand the force of water flow over the gills or skin, adaptive radiation has produced a wide array of opisthaptors in different species. Opisthaptors may bear large and small hooks, suckers, and clamps, often in combination with each other.
Common genera are Gyrodactylus (L. gyro, a circle, + Gr. daktylos, toe, finger) (Figure 14-16) and Dactylogyrus (Gr. daktylos, toe, finger, + L. gyro, a circle), both of economic importance to fish culturists, and Polystoma (Gr. polys, many, + stoma, mouth), found in the urinary bladder of frogs.
Class Cestoda
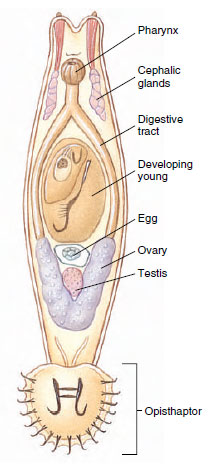
Cestoda, or tapeworms, differ in many respects from the preceding classes. They usually have long flat bodies in which there is a linear series of sets of reproductive organs. Each set is called a proglottid and usually has at its anterior and posterior ends zones of muscle weakness, marked externally by grooves. No digestive system is present. As in Monogenea and Trematoda, no external, motile cilia occur in adults, and the tegument is of a distal cytoplasm with sunken cell bodies beneath the superficial muscle layer (Figure 14-17). In contrast to monogeneans and trematodes, however, the entire surface of cestodes is covered with minute projections similar to microvilli of the vertebrate small intestine. These microtriches (sing microthrix) greatly enlarge the surface area of the tegument, which is a vital adaptation for a tapeworm since it must absorb all its nutrients across its tegument.
Tapeworms are nearly all monoecious. They have well-developed muscles, and their excretory system and nervous system are somewhat similar to those of other flatworms. They have no special sense organs but do have sensory endings in the tegument that are modified cilia (Figure 14-17). One of their most specialized structures is the scolex, or holdfast, which is the organ of attachment. It is usually provided with suckers or suckerlike organs and often with hooks or spiny tentacles (Figure 14-18).
 |
| Figure 14-17 Schematic drawing of a longitudinal section through a sensory ending in the tegument of Echinococcus granulosus. |
With rare exceptions, all cestodes require at least two hosts, and adults are parasites in the digestive tract of vertebrates. Often one of the intermediate hosts is an invertebrate.
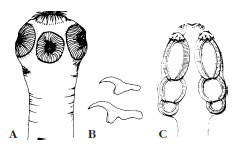 |
| Figure 14-18 Two tapeworm scolices. A, Scolex of Taenia solium (pork tapeworm) with apical hooks and suckers. (Scolex of Taenia saginata is similar, but without hooks.) B, Hooks of T. solium. C, Scolex of Acanthobothrium coronatum, a tapeworm of sharks. This species has largeleaflike sucker organs divided into chambers with apical suckers and hooks. |
Subclass Eucestoda contains the great majority of species in the class. With the exception of two small orders, members of this subclass have the body divided into a series of proglottids and are thus termed polyzoic. Larval forms of all eucestodes have six hooks. The main body of the worms, the chain of proglottids, is called a strobila. Typically, there is a germinative zone just behind the scolex where new proglottids are formed. As younger proglottids are differentiated in front of it, each individual proglottid moves posteriorly in the strobila, and its gonads mature. A proglottid is usually fertilized by another proglottid in the same or a different strobila. The shelled embryos form in the uterus of the proglottid, and either they are expelled through a uterine pore or the entire proglottid is shed from the worm as it reaches the posterior end.
Some zoologists have maintained that the proglottid formation of cestodes represents “true” segmentation (metamerism), but we do not support this view. Segmentation of tapeworms is best considered a replication of sex organs to increase reproductive capacity and is not related to the metamerism found in Annelida, Arthropoda, and Chordata.
More than 1000 species of tapeworms are known to parasitologists. Almost all vertebrate species are infected. Normally, adult tapeworms do little harm to their hosts. The most common tapeworms found in humans are given in Table 14-2.

Taenia saginata: Beef Tapeworm
Structure: Taenia saginata (Gr. tainia, band, ribbon) is called the beef tapeworm, but it lives as an adult in the alimentary canal of humans. Juvenile forms are found primarily in intermuscular tissue of cattle. A mature adult may reach a length of 10 m or more. Its scolex has four suckers for attachment to the intestinal wall, but no hooks. A short neck connects the scolex to the strobila, which may have as many as 2000 proglottids. Gravid proglottids bear shelled, infective larvae (Figure 14-19) and become detached and pass in feces.
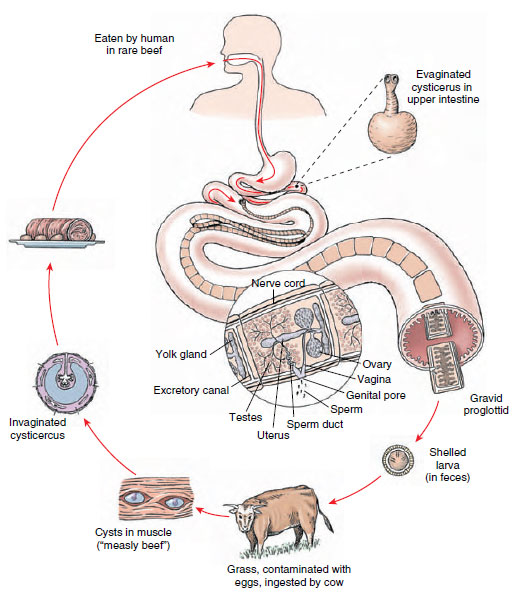 |
| Figure 14-19 Life cycle of beef tapeworm, Taenia saginata. Ripe proglottids break off in the human intestine, leave the body in feces, crawl out of feces onto grass, and are ingested by cattle. Eggs hatch in the cow’s intestine, freeing oncospheres, which penetrate into muscles and encyst, developing into “bladder worms.” A human eats infected rare beef, and cysticercus is freed in intestine where it attaches to the intestinal wall, forms a strobila, and matures. |
Tapeworms show some unity in their organization. Excretory canals in the scolex are also connected to the canals, two on each side, in the proglottids. Two longitudinal nerve cords from a nerve ring in the scolex run back into the proglottids (Figure 14-20). Attached to the excretory ducts are flame cells. Each mature proglottid also contains muscles and parenchyma as well as a complete set of male and female organs similar to those of a trematode.
In the order to which this species belongs, however, vitellaria are typically a single, compact vitelline gland located just posterior to the ovaries. When gravid proglottids break off and pass out with the feces, they usually crawl out of the fecal mass and onto vegetation nearby. There they may be picked up by grazing cattle. A proglottid ruptures as it dries, further scattering the embryos on soil and grass. Embryos may remain viable on grass for as long as 5 months.
Life Cycle: When cattle swallow shelled larvae, they hatch, and the larvae (oncospheres) use their hooks to burrow through the intestinal wall into blood or lymph vessels and finally reach voluntary muscle, where they encyst to become bladder worms (juveniles called cysticerci). There the juveniles develop an invaginated scolex but remain quiescent. When infected “measly” meat is eaten by a suitable host, the cyst wall dissolves, the scolex evaginates and attaches to the intestinal mucosa, and new proglottids begin to develop. It takes 2 to 3 weeks for a mature worm to form. When a person is infected with one of these tapeworms, numerous gravid proglottids are expelled daily, sometimes crawling out the anus by themselves. Humans become infected by eating rare roast beef, steaks, and barbecues. Considering that about 1% of American cattle are infected, that 20% of all cattle slaughtered are not federally inspected, and that even in inspected meat one-fourth of infections are missed, it is not surprising that tapeworm infection is fairly common. Infection is precluded when meat is thoroughly cooked.
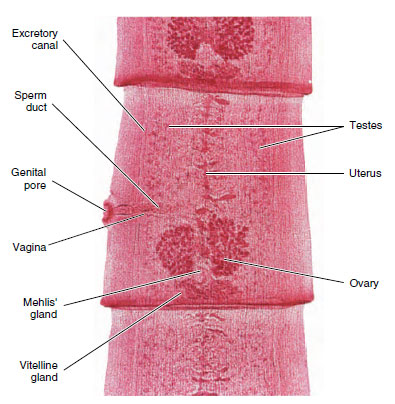 |
| Figure 14-20 Mature proglottid of Taenia pisiformis, a dog tapeworm. Portions of two other proglottids also shown. |
Some Other Tapeworms
Taenia solium: Pork Tapeworm Adult Taenia solium (Gr. tainia, band, ribbon) live in the small intestine of humans, whereas juveniles live in the muscles of pigs. The scolex has both suckers and hooks arranged on its tip (Figure 14-18), the rostellum. The life history of this worm is similar to that of the beef tapeworm, except that humans become infected by eating improperly cooked pork.
Taenia solium is much more dangerous than T. saginata because cysticerci, as well as adults, can develop in humans. If eggs or proglottids are accidentally ingested by a human, the liberated embryos migrate to any of several organs and form cysticerci (Figure 14-21). The condition is called cysticercosis. Common sites are the eye or brain, and infection in such locations can result in blindness, serious neurological symptoms, or death.
 |
| Figure 14-21 Section through the brain of a person who died of cerebral cysticercosis, an infection with cysticerci of Taenia solium. |
Diphyllobothrium latum: Fish Tapeworm Adult Diphyllobothrium (Gr. dis, double, + phyllon, leaf, + bothrion, hole, trench) are found in the intestine of humans, dogs, cats, and other mammals; immature stages are in crustaceans and fish. With a length of up to 20 m, it is the largest cestode that infects humans. Fish tapeworm infections can occur anywhere in the world where people commonly eat raw fish; in the United States infections are most common in the Great Lakes region. In Finland, but apparently not other areas, the worm can cause a serious anemia.
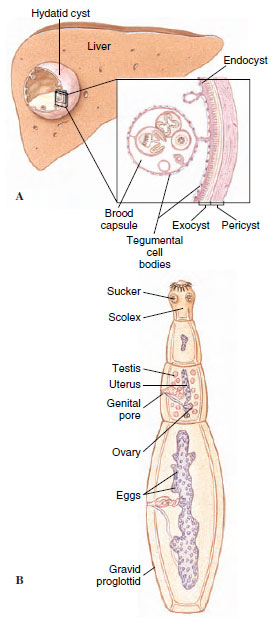 |
| Figure 14-22 Echinococcus granulosus, a dog tapeworm, which may be dangerous to humans. A, Early hydatid cyst or bladder-worm stage found in cattle, sheep, hogs, and sometimes humans produces hydatid disease. Humans acquire disease by unsanitary habits in association with dogs. When eggs are ingested, liberated larvae usually encyst in the liver, lungs, or other organs. Brood capsules containing scolices are formed from the inner layer of each cyst. The cyst enlarges, developing other cysts with brood pouches. It may grow for years to the size of a basketball, necessitating surgery. B, The adult tapeworm lives in intestine of a dog or other carnivore. |
Classification of Phylum Platyhelminthes
Class Turbellaria (tur´bel-lar´e-a) (L. turbellae [pl.], stir, bustle, + aria, like or connected with): turbellarians. Usually free-living forms with soft, flattened bodies; covered with ciliated epidermis containing secreting cells and rodlike bodies (rhabdites); mouth usually on ventral surface sometimes near center of body; no body cavity except intercellular lacunae in parenchyma; mostly hermaphroditic, but some have asexual fission. A paraphyletic taxon. Examples: Dugesia (planaria), Microstomum, Planocera.
Class Trematoda (trem´a-to´da) (Gr. trematodes, with holes, + eidos, form): digenetic flukes. Body of adults covered with a syncytial tegument without cilia; leaflike or cylindrical in shape; usually with oral and ventral suckers, no hooks; alimentary canal usually with two main branches; mostly monoecious; development indirect, with first host a mollusc, final host usually a vertebrate; parasitic in all classes of vertebrates. Examples: Fasciola, Clonorchis, Schistosoma.
Class Monogenea (mon´o-gen´e-a) (Gr. mono, single, + gene, origin, birth): monogenetic flukes. Body of adults covered with a syncytial tegument without cilia; body usually leaflike to cylindrical in shape; posterior attachment organ with hooks, suckers, or clamps, usually in combination; monoecious; development direct, with single host and usually with free-swimming, ciliated larva; all parasitic, mostly on skin or gills of fish. Examples: Dactylogyrus, Polystoma, Gyrodactylus.
Class Cestoda (ses-to´da) (Gr. kestos, girdle, + eidos, form): tapeworms. Body of adults covered with nonciliated, syncytial tegument; general form of body tapelike; scolex with suckers or hooks, sometimes both, for attachment; body usually divided into series of proglottids; no digestive organs; usually monoecious; larva with hooks; parasitic in digestive tract of all classes of vertebrates; development indirect with two or more hosts; first host may be vertebrate or invertebrate. Examples: Diphyllobothrium, Hymenolepis, Taenia.
Echinococcus granulosus: Unilocular Hydatid Adult E. granulosus (Gr. echinos, hedgehog, + kokkos, kernel) (Figure 14-22B), parasitizes dogs and other canines; juveniles develop in more than 40 species of mammals, including humans, monkeys, sheep, reindeer, and cattle. Thus humans may serve as an intermediate host in the case of this tapeworm. The juvenile stage is a special kind of cysticercus called a hydatid cyst (Gr. hydatis, watery vesicle). It grows slowly, but it can grow for a long time—up to 20 years—reaching the size of a basketball in an unrestricted site such as the liver. If the hydatid grows in a critical location, such as the heart or central nervous system, serious symptoms may appear in a much shorter time. The main cyst maintains a single or unilocular chamber, but within the main cyst, daughter cysts bud off, and each contains thousands of scolices. Each scolex will produce a worm when eaten by a canine. The only treatment is surgical removal of the hydatid.




