Nucleic Acids
Nucleic acids occur in two forms, deoxyribonucleic acid (DNA ) and ribonucleic acid (RNA). Both are linear, unbranched polymers of subunits termed nucleotides. DNA is found in the nucleus of eukaryotes and the cytoplasm or nucleoid of prokaryotes and functions as the molecule of heredity. RNA molecules are synthesized on DNA templates and participate in protein synthesis in the cytoplasm.Each nucleotide consists of three major parts: (1) a five-carbon sugar (pentose); (2) a flat, heterocyclic, nitrogen-containing organic base; and (3) a negatively charged phosphate group, which gives the polymer its acidic property (Figure 2-5). The nitrogenous base in each nucleotide is covalently attached to the sugar by a glycosidic bond. The phosphate group is also covalently linked to the sugar.
The sugar β-D-ribose is found in ribonucleotide monomers of RNA. The pentose in the deoxyribonucleotide monomers of DNA differ by the absence of oxygen at the #2 carbon and is thus 2-deoxy-β-D-ribose.
The organic bases are of two general types: single-ringed pyrimidines and double-ringed purines. The purines are adenine (A) and guanine (G). The pyrimidines are cytosine (C), thymine (T), and uracil (U). Thymine is found primarily in DNA and uracil is found only in RNA. In each polynucleotide strand of DNA and RNA, adjacent nucleotides are joined covalently by phosphodiester bonds between the 3' carbon of one nucleotide and the 5' carbon of the adjacent nucleotide.
Bases in the nucleotides spontaneously form hydrogen bonds in a highly specific manner. Adenine normally forms two hydrogen bonds with thymine in a complementary strand of the DNA double helix. Like wise, it can form two hydrogen bonds with U in DNA -RNA hybrids and in RNA-RNAinteractions. Guanine forms three hydrogen bonds with cytosine. DNA exists in the uniform shape of a double helix (Figure 2-6), with the complementary chains wound around each other like a spiral staircase, whereas RNA molecules are synthesized from DNA templates as single strands. The single strand of RNA, however, may fold back onto itself and form complementary base pairs to make unique secondary structures.
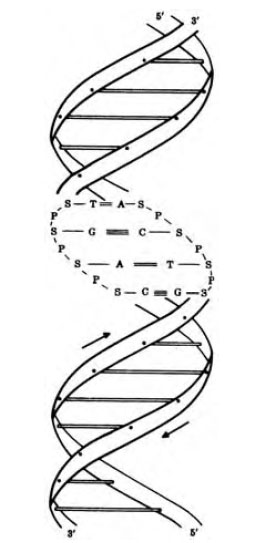 |
| Figure 2-6 Diagram of double helical DNA. |
The two complementary strands of a DNA double helix run in opposite directions, that is, they are antiparallel. If one chain is read from the 5' phosphate end, the other would read from the 3' hydroxyl. The double helix makes a turn every ten base pairs (approximately 3.4 nm). The paired bases are stacked in the center of the molecule, forming a hydrophobic core and giving the helix a width of about 2 nm.
There are three classes of RNA based on their functions: (1) transfer RNAs (tRNAs); (2) messenger RNAs (mRNAs); and (3) ribosomal RNAs (rRNAs). The tRNAs are the smallest (75-80 nucleotides in length) and serve to position each amino acid on the ribosome for polymerization into polypeptide chains. They contain a few unusual bases in addition to A, C, G, and U. The genetic code that specifies the amino acid sequences of proteins resides in the DNA sequence, and it becomes transcribed into complementary ribonucleotide sequences of mRNA, thus the length and composition of different mRNAs can vary greatly. The rRNAs are structural components of the ribosomes. There are three classes of rRNAs in bacteria and four in eukaryotes.
Notes
RNA has uracil in place of thymine.
Since A always pairs with T, and G always pairs with C, the purine:pyrimidine ratio in double stranded DNA is always 1.
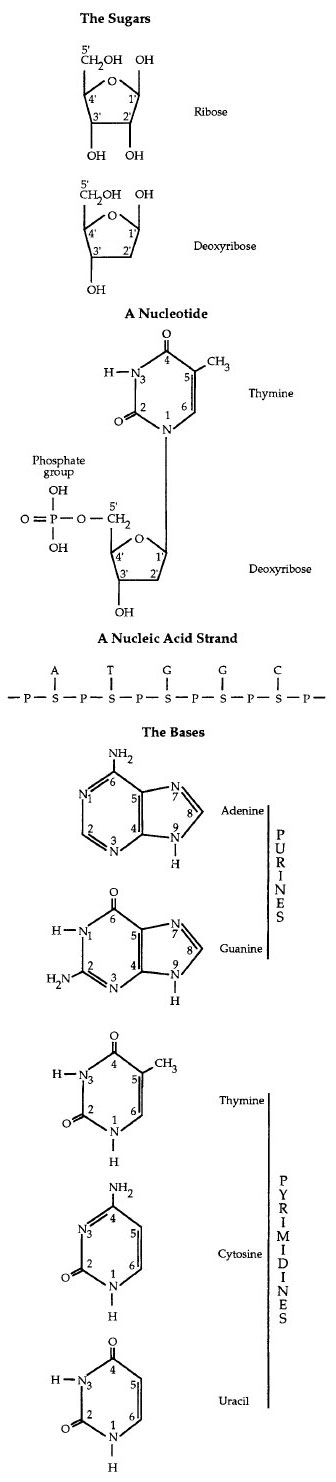
Figure 2-5 Structural components of nucleic acids




