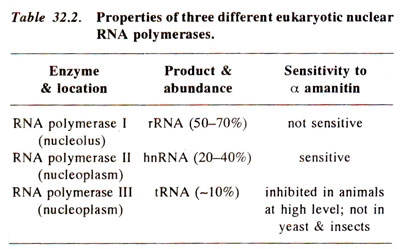
Multiple RNA polymerases in eukaryotes
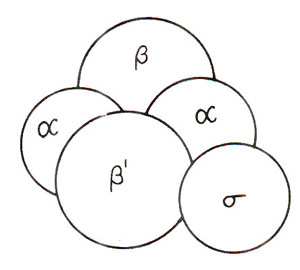
Fig. 32.2. A model of the structure of prokaryotic RNA polymerase showing association of five polypeptides (α2ββ').
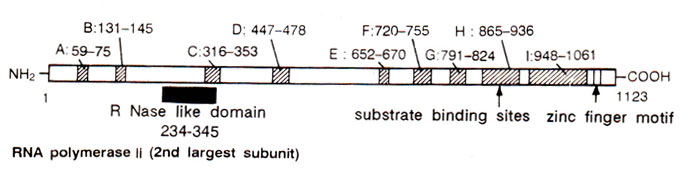
Fig. 32.12. Functional regions of the second largest subunit of RNA polymerase II of Drosophila. Solid bar shows similarity with barnase and other bacterial RNAses; A to I are regions having homology with prokaryotic RNA polymerase.
The three RNA polymerases can be distinguished on the basis of their sensitivity to certain inhibitors of transcription, e.g. α amanitin. Each of these enzymes is a large protein (~500,000 daltons), with two large and several (8 < 10) smaller subunits, some of them showing homology to the subunits of E. coll RNA polymerase. The largest subunit (~200,000 daltons) has homology with β', and the second large subunit (-140,000 daltons) has homology with β subunit of E. coli RNA polymerase (Fig. 32.2). One small subunit of RNA polymerase II also has homology with αsubunit. The second largest subunit of RNA polymerase II also shares the catalytic sites of ribonucleases, which may be used for RNA cleavage and may thus help in proof reading (Fig. 32.12).

Fig. 32.2. A model of the structure of prokaryotic RNA polymerase showing association of five polypeptides (α2ββ').

Fig. 32.12. Functional regions of the second largest subunit of RNA polymerase II of Drosophila. Solid bar shows similarity with barnase and other bacterial RNAses; A to I are regions having homology with prokaryotic RNA polymerase.