Subphylum Vertebrata (Craniata)
Subphylum
Vertebrata
(Craniata)
The third subphylum of the chordates is the large and diverse Vertebrata. This monophyletic group shares the basic chordate characteristics with the other two subphyla, but in addition it demonstrates a number of novel homologies that the others do not share. The alternative name of the subphylum, Craniata, more accurately describes the group since all have a cranium (bony or cartilaginous braincase) whereas, the jawless fishes lack vertebrae.
Adaptations that Have Guided Vertebrate Evolution
From the earliest fishes to the mammals, the evolution of the vertebrates has been guided by the specialized basic adaptations of living endoskeleton, pharynx and efficient respiration, advanced nervous system, and paired limbs.
Living Endoskeleton
The endoskeleton of vertebrates, as in the echinoderms, is an internal supportive structure and framework for the body. This internal location is a departure in animal architecture, since invertebrate skeletons generally enfold the body. Exoskeletons and endoskeletons have their own particular sets of advantages and limitations that are related to size. For vertebrates, the living endoskeleton possesses an overriding advantage over the dead exoskeleton of arthropods. Growing with the body as it does, the endoskeleton permits almost unlimited body size with much greater economy of building materials. Some vertebrates have become the most massive animals on earth. The endoskeleton forms an excellent jointed scaffolding for muscles and the muscles in turn protect the skeleton and cushion it from potentially damaging impact.
We should note that vertebrates have not wholly lost the protective function of a firm external covering. The skull and thoracic rib cage enclose and protect vulnerable organs. Most vertebrates are further protected with a tough integument, often bearing nonliving structures such as scales, hair, or feathers that may provide insulation as well as physical security.
The endoskeleton was probably composed initially of cartilage that later gave way to bone. Cartilage forms a perfectly suitable endoskeleton for aquatic animals. Cartilage is superior to bone for fast growth and is therefore ideal for constructing the first skeletal framework of all vertebrate embryos. In agnathans (hagfish and lampreys), sharks and their kin, and even in some bony fishes such as sturgeons, the adult endoskeleton is composed mostly or entirely of cartilage. Bone appears in the endoskeleton of more derived vertebrates, perhaps because it offers two clear advantages to cartilage. First, it serves as a reservoir for phosphate, an indispensable component of compounds with high-energy bonds, of membranes, and of nucleic acids. Second, only bone could provide the structural strength required for life on land, where mechanical stresses on the endoskeleton are far greater than they are in water.
Pharynx and Efficient Respiration
The perforated pharynx, present as pharyngeal pouches in all chordates at some stage in their life cycle, evolved for filter-feeding. In primitive chordates (such as amphioxus), water with suspended food particles is drawn through the mouth by ciliary action and flows out through the pharyngeal slits where food is trapped in mucus. As protovertebrates shifted from filter-feeding to a predatory life habit, the pharynx became modified into a muscular feeding apparatus through which water could be pumped by expanding and contracting the pharyngeal cavity. Circulation to the internal gills was improved by addition of capillary beds (lacking in protochordates) and development of a ventral heart and muscular aortic arches. All of these changes supported an increased metabolic rate that would have to accompany the switch to an active life of selective predation.
New Head and Advanced Nervous System
No single system in the body is more strongly associated with functional and structural advancement than is the nervous system. When vertebrate ancestors shifted from filter feeding to active predation, new sensory, motor, and integrative controls became essential for location and capture of larger prey items. Paired special sense organs designed for distance reception evolved. These included paired eyes with lenses and inverted retinas; pressure receptors, such as paired ears designed for equilibrium and later redesigned to include sound reception; electroreceptors that could signal the direction of potential prey; and chemical receptors, including taste receptors and exquisitely sensitive olfactory organs.
Development of the vertebrate head and paired sense organs was largely the result of two embryonic innovations present only in vertebrates: the neural crest and epidermal placodes. The neural crest, a population of ectodermal cells lying along the length of the embryonic neural tube, contributes to the formation of many different structures, among them the cranium, cranial nerves, branchial skeleton, and the aortic arches. The epidermal placodes are plate-like ectodermal thickenings (the term “placode” derives from a Greek word meaning “plate”) that appear anteriorly on either side of the neural tube. These give rise to the nose, eyes, ears, taste receptors, and lateral line mechanoreceptors and electroreceptors. Thus the vertebrate head with its sensory structures located adjacent to the mouth (later equipped with prey-capturing jaws), stemmed from the creation of completely new cell types— a rare event in animal evolution.
Paired Limbs
Pectoral and pelvic appendages are present in most vertebrates in the form of paired fins or jointed legs. These originated as swimming stabilizers and later became prominently developed into legs for locomotion on land. Jointed limbs are especially suited for life on land because they permit finely graded leveling motions against a substrate.
Characteristics of Subphylum Vertebrata
The Search for the Vertebrate Ancestral Stock
The earliest vertebrate Paleozoic fossils,
the jawless ostracoderm fishes we consider
at the end of this section, share
many novel features or organ system
development with living vertebrates.
These organ systems therefore must
have originated in either an early vertebrate
or invertebrate chordate lineage.
With one exception, hardly any invertebrate
chordates are known as fossils.
The exception is Pikaia gracilens, a ribbon-
shaped, somewhat fishlike, creature
about 5 cm in length discovered in
the famous Burgess Shale of British
Columbia (Figure 25-11). Pikaia is a
mid-Cambrian form that precedes the
earliest vertebrate fossils by many millions
of years. This fossil possessed both a notochord and characteristic
chordate>-shaped muscle bands
(myotomes). Without question Pikaia is
a chordate. It shows a remarkable
resemblance to living amphioxus, at
least in overall body organization, and
may in fact be an early cephalochordate. Pikaia, and a slightly older similar
fossil recently discovered in China,
called Yunnanozoon, are provocative
fossils but, until other Cambrian chordate
fossils are discovered their relationship
to earliest vertebrates remains
uncertain. In the absence of additional
fossil evidence, most speculations on
vertebrate ancestry have focused on the
living cephalochordates and tunicates,
since it is widely believed that vertebrates
must have emerged from a lineage
resembling one of these protochordate
groups.
Garstang’s Hypothesis of Chordate Larval Evolution
At first glance, tunicates seem unlikely candidates as ancestors for vertebrates. The adult tunicate, which spends it life anchored to some marine surface, lacks a notochord, tubular nerve cord, postanal tail, sense organs, and segmented musculature. Its larva, however, bears all the right qualifications for chordate membership. Called “tadpole” larva because of its superficial resemblance to larval frogs, this tiny, site-seeking form has a notochord, hollow dorsal nerve cord, pharyngeal slits, and postanal tail, as well as a brain and sense organs.
At the time of its discovery in 1869, the tadpole larva was considered a descendant of an ancient free-swimming chordate ancestor of tunicates. The adults then were regarded as degenerate, sessile descendants of the freeswimming form. In 1928, Walter Garstang in England introduced fresh thinking into the vertebrate ancestor debate by turning this sequence around; rather than the ancestral tadpole larva giving rise to a degenerative tunicate sessile adult, he suggested that the sessile adults were the ancestral stock. The tadpole larva then evolved as an adaptation for spreading to new habitats. Next, Garstang suggested that at some point the tadpole larva failed to metamorphose into an adult, but developed gonads and reproduced in the larval stage. With continued larval evolution, a new group of free-swimming animals appeared (Figure 25-12).
Garstang called this process paedomorphosis (Gr. pais, child + morphe, form), a term describing the evolutionary retention of juvenile or larval traits in the adult body. Garstang departed from previous thinking by suggesting that evolution may occur in larval stages of animals—and in this case, lead to the vertebrate lineage. Paedomorphosis is a well-known phenomenon in several different animal groups. Furthermore, Garstang’s hypothesis agrees with the embryological evidence. Nevertheless, it remains untested and speculative.
Position of Amphioxus
For many years zoologists believed that the cephalochordate amphioxus is the closest living relative of vertebrates. No other protochordate shows the basic diagnostic characteristics of the chordates so well. However, as pointed out in the prologue to this section, amphioxus is no longer considered a direct ancestor of the vertebrates, although it may closely resemble an ancestor of the vertebrate lineage. It lacks a brain and all of the specialized sensory equipment that characterizes vertebrates. There are no gills in the pharynx and no mouth or pharyngeal musculature for pumping water through the gill slits; movement of water is entirely by the action of cilia.
Recent studies of the expression of homeobox-containing genes which control the body plan of chordate embryos suggest that the ancestor of both amphioxus and vertebrates was cephalized; it had a head region with a brain and sense organs. In amphioxus and other cephalochordates the notochord grows forward to the anterior tip of the animal, obliterating most traces of the primitive head region. Despite these specializations and others peculiar to modern cephalochordates, many zoologists believe that amphioxus has largely retained the primitive pattern of the immediate prevertebrate condition. Thus cephalochordates are probably the sister group of vertebrates (Figure 25-3).
The Ammocoete Larva of Lampreys as a Model of the Primitive Vertebrate Body Plan
Lampreys (jawless fishes of the class Cephalaspidomorphi, discussed in the Fishes) have a freshwater larval stage known as the ammocoete (Figure 25-13). In body form, appearance, life habit, and most anatomical details, the ammocoete larva resembles amphioxus. In fact, lamprey larvae were given the genus name Ammocoetes (Gr. ammos, sand, + koite, bed, referring to the preferred larval habitat) in the nineteenth century when it was erroneously thought to be an adult cephalochordate, closely allied with amphioxus. Ammocoete larvae are so different from adult lampreys that the mistake is understandable; the exact relationship was not explained until metamorphosis into the adult lamprey was observed.
Ammocoete larvae have a long, slender body with an oral hood surrounding the mouth much like amphioxus (Figure 25-13). Ammocoetes are filter feeders, but instead of drawing water by ciliary action into the pharynx as amphioxus does, ammocoetes produce a feeding current by muscular pumping action much like modern fishes. In the floor of the pharynx is an endostyle, as in amphioxus. The endostyle produces a food-ensnaring mucus that is passed directly to the intestine. The arrangement of body muscle into myotomes, the presence of a notochord serving as chief skeletal axis, and the plan of the circulatory system all closely resemble these features in amphioxus.
Ammocoetes do have several characteristics lacking in amphioxus that are homologous to those of vertebrates. These include a two-chambered heart (atrium and ventricle), a three-part brain (forebrain, midbrain, hindbrain), special sense organs derived from epidermal placodes (two eyes, one on each side of the midbrain; a median nostril; and auditory vesicles located lateral to the midbrain), a thyroid gland, and a pituitary gland. The kidney is pronephric and conforms to the basic vertebrate plan. Instead of the numerous pharyngeal slits of amphioxus, there are only seven pairs of pharyngeal pouches and slits in ammocoetes. From pharyngeal bars separating the pharyngeal slits project gill filaments bearing secondary lamellae much like the more extensive gills of modern fishes (see Figure 26-28). Ammocoetes also have a true liver replacing the hepatic cecum of amphioxus, a gallbladder, and pancreatic tissue (but no distinct pancreatic gland).
Ammocoete larvae display the most primitive condition for these characteristics of any living vertebrate. It clearly illustrates many shared derived characters of vertebrates that are obscured in the development of other vertebrates. It may approach most closely the supposed body plan of the ancestral vertebrate.
The Earliest Vertebrates: Jawless Ostracoderms
The earliest vertebrate fossils are late Cambrian articulated skeletons from the United States, Bolivia, and Australia. They were small, jawless creatures collectively called ostracoderms (os-trak´o-derm) (Gr. ostrakon, shell, + derma, skin), which belong to the Agnatha division of the vertebrates. These earliest ostracoderms lacked paired fins that later fishes found so important for stability (Figure 25-14). The swimming movements of one of the early groups, the heterostracans (Gr. heteros, different, + ostrakon, shell) (also called pteraspidiforms), must have been clumsy, although sufficient to propel them along the ocean bottom where they searched for food. With fixed circular or slitlike mouth openings they probably filtered small food particles from the water or ocean bottom. However, unlike the ciliary filter-feeding protochordates, ostracoderms sucked water into the pharynx by muscular pumping, an important innovation that suggests to some authorities that ostracoderms may have been mobile predators that fed on softbodied animals.
During the Devonian period, the heterostracans underwent a major radiation, resulting in the appearance of several peculiar-looking forms varying in shape and length of the snout, dorsal spines, and dermal plates. Without ever evolving paired fins or jaws, these earliest vertebrates flourished for 150 million years until becoming extinct near the end of the Devonian period.
Coexisting with heterostracans throughout much of the Devonian period were osteostracans (Gr. osteon, bone, + ostrakon, shell) (also called cephalaspidiforms). Osteostracans improved the efficiency of their benthic life by evolving paired pectoral fins that provided control over pitch and yaw. This innovation ensured well-directed forward movement. A typical osteostracan, such as Cephalaspis (Gr. kephale, head, + aspis, shield) (Figure 25-14), was a small animal, seldom exceeding 30 cm in length. It was covered by a welldeveloped armor—the head by a solid shield and the body by bony plates— but it had no axial skeleton or vertebrae. Their jawless mouth was toothless. Other distinctive features included a sensory lateral line system, paired eyes with complex eye muscle patterns, and inner ears with semicircular canals.
Another group of ostracoderms, the anaspids, (Figure 25-14) were more streamlined and more closely resembled modern-day jawless fishes (lamprey, for example) than any other ostracoderm. The evolution of the basic vertebrate head pattern in ostracoderms, although lacking jaws, was an advance of great significance in vertebrate history. As a group the bottom-feeding ostracoderms enjoyed a respectable radiation in the Silurian and Devonian periods.
For decades, geologists have used strange microscopic, toothlike fossils called conodonts (Gr. konos, cone, + odontos, tooth) to date Paleozoic marine sediments without having any idea what kind of creature originally possessed these elements. The discovery in the early 1980s of fossils of complete conodont animals has changed this situation: conodont elements belonged to a small early marine vertebrate (Figure 25-15). It is widely believed that as more is learned about conodont animals they will play an important role in understanding the origin of vertebrates. At present, however, their position in vertebrate phylogeny is a matter of debate.
Early Jawed Vertebrates
All jawed vertebrates, whether extinct or living, are collectively called gnathostomes (“jaw mouth”) in contrast to the jawless vertebrates, the agnathans (“without jaw”). Living agnathans, the naked hagfishes and lampreys, also are often called cyclostomes (“circle mouth”). The gnathostomes are a monophyletic group since presence of jaws is a derived character state shared by all jawed fishes and tetrapods. Agnathans, however, are defined principally by the absence of a feature—jaws—that characterize the gnathostomes. Therefore the superclass Agnatha may be paraphyletic.
The origin of jaws was one of the most important events in vertebrate evolution. The utility of jaws is obvious: they allow predation on large and active forms of food not available to jawless vertebrates. Ample evidence suggests that jaws arose through modifications of the first two of the serially repeated cartilaginous gill arches. We can see the beginnings of this trend in some ostracoderms where the mouth becomes bordered by strong dermal plates that could be manipulated somewhat like jaws with the gill arch musculature. Later, the anterior gill arches became hinged and bent forward into the characteristic position of vertebrate jaws. Evidence for this remarkable transformation is threefold. First, both gill arches and jaws form from upper and lower bars that bend forward and are hinged in the middle (Figure 25-16). Second, both gill arches and jaws are derived from neural crest cells rather than from mesodermal tissue, the source of most bones. Third, the jaw musculature is homologous to the original gill support musculature. Nearly as remarkable as this drastic morphological remodeling is the subsequent evolutionary fate of jawbone elements—their transformation into ear ossicles of the mammalian middle ear.
Among the first jawed vertebrates were the heavily armored placoderms (plak´cute;avo-derm) (Gr. plax, plate, + derma, skin). These first appear in the fossil record in the early Devonian period (Figure 25-17). Placoderms evolved a great variety of forms, some very large (one was 10 m in length!) and grotesque in appearance. They were armored fish covered with diamondshaped scales or with large plates of bone. All became extinct by the end of the Paleozoic era and appear to have left no descendants. However, the acanthodians (Figure 25-17), a group of early jawed fishes that were contemporary with the placoderms, may have given rise to the great radiation of bony fishes that dominate the waters of the world today.
Evolution of Modern Fishes and Tetrapods
Reconstruction of the origins of the vast and varied assemblage of modern living vertebrates is based, as we have seen, largely on fossil evidence. Unfortunately the fossil evidence for the earliest vertebrates is often incomplete and tells us much less than we would like to know about subsequent trends in evolution. Affinities become much easier to establish as the fossil record improves. For instance, the descent of birds and mammals from early tetrapod ancestors has been worked out in a highly convincing manner from the relatively abundant fossil record available. By contrast, the ancestry of modern fishes is shrouded in uncertainty.
Despite the difficulty of clarifying early lines of descent for vertebrates, they are clearly a natural, monophyletic group, distinguished by a large number of shared characteristics. We still do not know, however, from which chordate group the vertebrate lineage originated. Early in their evolution, vertebrates divided into agnathans and gnathostomes. These two groups differ from each other in many fundamental ways, in addition to absence of jaws in the former group and their presence in the latter. The appearance of both jaws and paired fins were major innovations in vertebrate evolution, among the most important reasons for the subsequent major radiations of vertebrates that produced the modern fishes and all of the tetrapods, including you, the reader of this section.
Traditional Linnean
Classification of Living Members of Phylum Chordata
Phylum Chordata
The third subphylum of the chordates is the large and diverse Vertebrata. This monophyletic group shares the basic chordate characteristics with the other two subphyla, but in addition it demonstrates a number of novel homologies that the others do not share. The alternative name of the subphylum, Craniata, more accurately describes the group since all have a cranium (bony or cartilaginous braincase) whereas, the jawless fishes lack vertebrae.
Adaptations that Have Guided Vertebrate Evolution
From the earliest fishes to the mammals, the evolution of the vertebrates has been guided by the specialized basic adaptations of living endoskeleton, pharynx and efficient respiration, advanced nervous system, and paired limbs.
Living Endoskeleton
The endoskeleton of vertebrates, as in the echinoderms, is an internal supportive structure and framework for the body. This internal location is a departure in animal architecture, since invertebrate skeletons generally enfold the body. Exoskeletons and endoskeletons have their own particular sets of advantages and limitations that are related to size. For vertebrates, the living endoskeleton possesses an overriding advantage over the dead exoskeleton of arthropods. Growing with the body as it does, the endoskeleton permits almost unlimited body size with much greater economy of building materials. Some vertebrates have become the most massive animals on earth. The endoskeleton forms an excellent jointed scaffolding for muscles and the muscles in turn protect the skeleton and cushion it from potentially damaging impact.
We should note that vertebrates have not wholly lost the protective function of a firm external covering. The skull and thoracic rib cage enclose and protect vulnerable organs. Most vertebrates are further protected with a tough integument, often bearing nonliving structures such as scales, hair, or feathers that may provide insulation as well as physical security.
The endoskeleton was probably composed initially of cartilage that later gave way to bone. Cartilage forms a perfectly suitable endoskeleton for aquatic animals. Cartilage is superior to bone for fast growth and is therefore ideal for constructing the first skeletal framework of all vertebrate embryos. In agnathans (hagfish and lampreys), sharks and their kin, and even in some bony fishes such as sturgeons, the adult endoskeleton is composed mostly or entirely of cartilage. Bone appears in the endoskeleton of more derived vertebrates, perhaps because it offers two clear advantages to cartilage. First, it serves as a reservoir for phosphate, an indispensable component of compounds with high-energy bonds, of membranes, and of nucleic acids. Second, only bone could provide the structural strength required for life on land, where mechanical stresses on the endoskeleton are far greater than they are in water.
Pharynx and Efficient Respiration
The perforated pharynx, present as pharyngeal pouches in all chordates at some stage in their life cycle, evolved for filter-feeding. In primitive chordates (such as amphioxus), water with suspended food particles is drawn through the mouth by ciliary action and flows out through the pharyngeal slits where food is trapped in mucus. As protovertebrates shifted from filter-feeding to a predatory life habit, the pharynx became modified into a muscular feeding apparatus through which water could be pumped by expanding and contracting the pharyngeal cavity. Circulation to the internal gills was improved by addition of capillary beds (lacking in protochordates) and development of a ventral heart and muscular aortic arches. All of these changes supported an increased metabolic rate that would have to accompany the switch to an active life of selective predation.
New Head and Advanced Nervous System
No single system in the body is more strongly associated with functional and structural advancement than is the nervous system. When vertebrate ancestors shifted from filter feeding to active predation, new sensory, motor, and integrative controls became essential for location and capture of larger prey items. Paired special sense organs designed for distance reception evolved. These included paired eyes with lenses and inverted retinas; pressure receptors, such as paired ears designed for equilibrium and later redesigned to include sound reception; electroreceptors that could signal the direction of potential prey; and chemical receptors, including taste receptors and exquisitely sensitive olfactory organs.
Development of the vertebrate head and paired sense organs was largely the result of two embryonic innovations present only in vertebrates: the neural crest and epidermal placodes. The neural crest, a population of ectodermal cells lying along the length of the embryonic neural tube, contributes to the formation of many different structures, among them the cranium, cranial nerves, branchial skeleton, and the aortic arches. The epidermal placodes are plate-like ectodermal thickenings (the term “placode” derives from a Greek word meaning “plate”) that appear anteriorly on either side of the neural tube. These give rise to the nose, eyes, ears, taste receptors, and lateral line mechanoreceptors and electroreceptors. Thus the vertebrate head with its sensory structures located adjacent to the mouth (later equipped with prey-capturing jaws), stemmed from the creation of completely new cell types— a rare event in animal evolution.
Paired Limbs
Pectoral and pelvic appendages are present in most vertebrates in the form of paired fins or jointed legs. These originated as swimming stabilizers and later became prominently developed into legs for locomotion on land. Jointed limbs are especially suited for life on land because they permit finely graded leveling motions against a substrate.
Characteristics of Subphylum Vertebrata
- Chief diagnostic features of chordates—notochord, dorsal nerve cord, pharyngeal pouches, and postanal tail—all present at some stage of the life cycle
- Integument basically of two divisions, an outer epidermis of stratified epithelium from ectoderm and an inner dermis of connective tissue derived from mesoderm; many modifications of skin among the various classes, such as glands, scales, feathers, claws, horns, and hair
- Distinctive endoskeleton consisting of vertebral column (notochord persistent in jawless fishes which lack vertebrae), limb girdles, and two pairs of jointed appendages derived from somatic mesoderm, and a head skeleton (cranium and pharyngeal skeleton) derived largely from neural crest cells
- Muscular, perforated pharynx; in fishes pharyngeal slits possess gills and muscular aortic arches; in tetrapods the much reduced pharynx is embryonic source of glandular tissue
- Many muscles attached to the skeleton to provide for movement
- Complete digestive system ventral to the spinal column and provided with large digestive glands, liver, and pancreas
- Circulatory system consisting of a ventral heart of two to four chambers; closed blood vessel system of arteries, veins, and capillaries; blood fluid containing red blood corpuscles with hemoglobin and white corpuscles; paired aortic arches connecting ventral and dorsal aortas and giving off branches to the gills among gillbreathing vertebrates; in terrestrial types modification of the aortic arch plan into pulmonary and systemic systems
- Well-developed coelom largely filled with the visceral systems
- Excretory system consisting of paired kidneys (mesonephric or metanephric types in adults) provided with ducts to drain the waste to cloaca or anal region
- Highly differentiated brain; 10 or 12 pairs of cranial nerves with both motor and sensory functions usually; a pair of spinal nerves for each primitive myotome; an autonomic nervous system in control of involuntary functions of internal organs; paired special sense organs derived from epidermal placodes
- Endocrine system of ductless glands scattered through the body
- Nearly always separate sexes; each sex containing paired gonads with ducts that discharge their products either into the cloaca or into special openings near the anus
- Body plan consisting typically of head, trunk, and postanal tail; neck present in some, especially terrestrial forms; two pairs of appendages usually, although entirely absent in some; coelom divided into a pericardial space and a general body cavity; mammals with a thoracic cavity
The Search for the Vertebrate Ancestral Stock
 |
| Figure 25-11 Pikaia, the earliest known chordate, from the Burgess Shale of British Columbia, Canada. |
Garstang’s Hypothesis of Chordate Larval Evolution
At first glance, tunicates seem unlikely candidates as ancestors for vertebrates. The adult tunicate, which spends it life anchored to some marine surface, lacks a notochord, tubular nerve cord, postanal tail, sense organs, and segmented musculature. Its larva, however, bears all the right qualifications for chordate membership. Called “tadpole” larva because of its superficial resemblance to larval frogs, this tiny, site-seeking form has a notochord, hollow dorsal nerve cord, pharyngeal slits, and postanal tail, as well as a brain and sense organs.
At the time of its discovery in 1869, the tadpole larva was considered a descendant of an ancient free-swimming chordate ancestor of tunicates. The adults then were regarded as degenerate, sessile descendants of the freeswimming form. In 1928, Walter Garstang in England introduced fresh thinking into the vertebrate ancestor debate by turning this sequence around; rather than the ancestral tadpole larva giving rise to a degenerative tunicate sessile adult, he suggested that the sessile adults were the ancestral stock. The tadpole larva then evolved as an adaptation for spreading to new habitats. Next, Garstang suggested that at some point the tadpole larva failed to metamorphose into an adult, but developed gonads and reproduced in the larval stage. With continued larval evolution, a new group of free-swimming animals appeared (Figure 25-12).
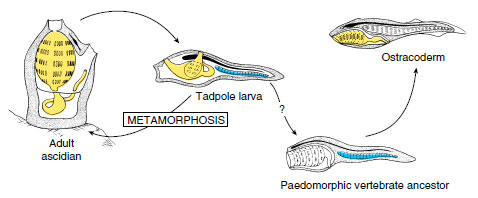 |
| Figure 25-12 Garstang’s hypothesis of larval evolution. Adult tunicates live on the sea floor but produce a free swimming tadpole larva. More than 500 million years ago, some larvae began to reproduce in the swimming stage. These evolved into ostracoderms, the first known vertebrates. |
Garstang called this process paedomorphosis (Gr. pais, child + morphe, form), a term describing the evolutionary retention of juvenile or larval traits in the adult body. Garstang departed from previous thinking by suggesting that evolution may occur in larval stages of animals—and in this case, lead to the vertebrate lineage. Paedomorphosis is a well-known phenomenon in several different animal groups. Furthermore, Garstang’s hypothesis agrees with the embryological evidence. Nevertheless, it remains untested and speculative.
Position of Amphioxus
For many years zoologists believed that the cephalochordate amphioxus is the closest living relative of vertebrates. No other protochordate shows the basic diagnostic characteristics of the chordates so well. However, as pointed out in the prologue to this section, amphioxus is no longer considered a direct ancestor of the vertebrates, although it may closely resemble an ancestor of the vertebrate lineage. It lacks a brain and all of the specialized sensory equipment that characterizes vertebrates. There are no gills in the pharynx and no mouth or pharyngeal musculature for pumping water through the gill slits; movement of water is entirely by the action of cilia.
Recent studies of the expression of homeobox-containing genes which control the body plan of chordate embryos suggest that the ancestor of both amphioxus and vertebrates was cephalized; it had a head region with a brain and sense organs. In amphioxus and other cephalochordates the notochord grows forward to the anterior tip of the animal, obliterating most traces of the primitive head region. Despite these specializations and others peculiar to modern cephalochordates, many zoologists believe that amphioxus has largely retained the primitive pattern of the immediate prevertebrate condition. Thus cephalochordates are probably the sister group of vertebrates (Figure 25-3).
The Ammocoete Larva of Lampreys as a Model of the Primitive Vertebrate Body Plan
Lampreys (jawless fishes of the class Cephalaspidomorphi, discussed in the Fishes) have a freshwater larval stage known as the ammocoete (Figure 25-13). In body form, appearance, life habit, and most anatomical details, the ammocoete larva resembles amphioxus. In fact, lamprey larvae were given the genus name Ammocoetes (Gr. ammos, sand, + koite, bed, referring to the preferred larval habitat) in the nineteenth century when it was erroneously thought to be an adult cephalochordate, closely allied with amphioxus. Ammocoete larvae are so different from adult lampreys that the mistake is understandable; the exact relationship was not explained until metamorphosis into the adult lamprey was observed.
Ammocoete larvae have a long, slender body with an oral hood surrounding the mouth much like amphioxus (Figure 25-13). Ammocoetes are filter feeders, but instead of drawing water by ciliary action into the pharynx as amphioxus does, ammocoetes produce a feeding current by muscular pumping action much like modern fishes. In the floor of the pharynx is an endostyle, as in amphioxus. The endostyle produces a food-ensnaring mucus that is passed directly to the intestine. The arrangement of body muscle into myotomes, the presence of a notochord serving as chief skeletal axis, and the plan of the circulatory system all closely resemble these features in amphioxus.
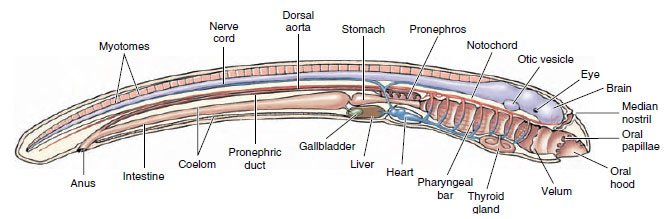 |
| Figure 25-13 Ammocoete larva, freshwater larval stage of a sea lamprey. Although they resemble amphioxus in many ways, ammocoetes have a well-developed brain, paired eyes, pronephric kidney, and other features lacking in amphioxus but representative of the vertebrate body plan. |
Ammocoetes do have several characteristics lacking in amphioxus that are homologous to those of vertebrates. These include a two-chambered heart (atrium and ventricle), a three-part brain (forebrain, midbrain, hindbrain), special sense organs derived from epidermal placodes (two eyes, one on each side of the midbrain; a median nostril; and auditory vesicles located lateral to the midbrain), a thyroid gland, and a pituitary gland. The kidney is pronephric and conforms to the basic vertebrate plan. Instead of the numerous pharyngeal slits of amphioxus, there are only seven pairs of pharyngeal pouches and slits in ammocoetes. From pharyngeal bars separating the pharyngeal slits project gill filaments bearing secondary lamellae much like the more extensive gills of modern fishes (see Figure 26-28). Ammocoetes also have a true liver replacing the hepatic cecum of amphioxus, a gallbladder, and pancreatic tissue (but no distinct pancreatic gland).
Ammocoete larvae display the most primitive condition for these characteristics of any living vertebrate. It clearly illustrates many shared derived characters of vertebrates that are obscured in the development of other vertebrates. It may approach most closely the supposed body plan of the ancestral vertebrate.
The Earliest Vertebrates: Jawless Ostracoderms
The earliest vertebrate fossils are late Cambrian articulated skeletons from the United States, Bolivia, and Australia. They were small, jawless creatures collectively called ostracoderms (os-trak´o-derm) (Gr. ostrakon, shell, + derma, skin), which belong to the Agnatha division of the vertebrates. These earliest ostracoderms lacked paired fins that later fishes found so important for stability (Figure 25-14). The swimming movements of one of the early groups, the heterostracans (Gr. heteros, different, + ostrakon, shell) (also called pteraspidiforms), must have been clumsy, although sufficient to propel them along the ocean bottom where they searched for food. With fixed circular or slitlike mouth openings they probably filtered small food particles from the water or ocean bottom. However, unlike the ciliary filter-feeding protochordates, ostracoderms sucked water into the pharynx by muscular pumping, an important innovation that suggests to some authorities that ostracoderms may have been mobile predators that fed on softbodied animals.
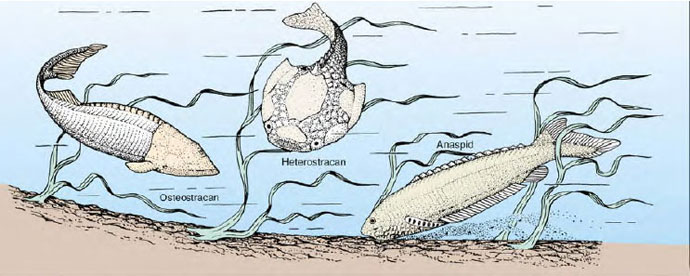 |
| Figure 25-14 Three ostracoderms, jawless fishes of Silurian and Devonian times. They are shown as they might have appeared while searching for food on the floor of a Devonian sea. All were probably filter-feeders, but employed a strong pharyngeal pump to circulate water rather than the much more limiting mode of ciliary feeding used by their protovertebrate ancestors (presumably resembling amphioxus for this feature). Modern lampreys are believed to be derived from the anaspid group. |
During the Devonian period, the heterostracans underwent a major radiation, resulting in the appearance of several peculiar-looking forms varying in shape and length of the snout, dorsal spines, and dermal plates. Without ever evolving paired fins or jaws, these earliest vertebrates flourished for 150 million years until becoming extinct near the end of the Devonian period.
Coexisting with heterostracans throughout much of the Devonian period were osteostracans (Gr. osteon, bone, + ostrakon, shell) (also called cephalaspidiforms). Osteostracans improved the efficiency of their benthic life by evolving paired pectoral fins that provided control over pitch and yaw. This innovation ensured well-directed forward movement. A typical osteostracan, such as Cephalaspis (Gr. kephale, head, + aspis, shield) (Figure 25-14), was a small animal, seldom exceeding 30 cm in length. It was covered by a welldeveloped armor—the head by a solid shield and the body by bony plates— but it had no axial skeleton or vertebrae. Their jawless mouth was toothless. Other distinctive features included a sensory lateral line system, paired eyes with complex eye muscle patterns, and inner ears with semicircular canals.
Another group of ostracoderms, the anaspids, (Figure 25-14) were more streamlined and more closely resembled modern-day jawless fishes (lamprey, for example) than any other ostracoderm. The evolution of the basic vertebrate head pattern in ostracoderms, although lacking jaws, was an advance of great significance in vertebrate history. As a group the bottom-feeding ostracoderms enjoyed a respectable radiation in the Silurian and Devonian periods.
For decades, geologists have used strange microscopic, toothlike fossils called conodonts (Gr. konos, cone, + odontos, tooth) to date Paleozoic marine sediments without having any idea what kind of creature originally possessed these elements. The discovery in the early 1980s of fossils of complete conodont animals has changed this situation: conodont elements belonged to a small early marine vertebrate (Figure 25-15). It is widely believed that as more is learned about conodont animals they will play an important role in understanding the origin of vertebrates. At present, however, their position in vertebrate phylogeny is a matter of debate.
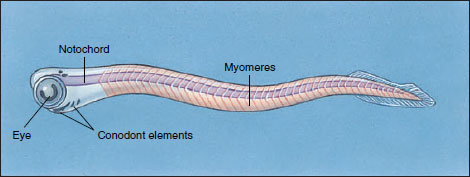 |
| Figure 25-15 Restoration of a living conodont animal. Conodonts superficially resembled amphioxus, but they possessed a much greater degree of encephalization (large, paired eyes, possible auditory capsules) and bonelike mineralized elements—all indicating that conodont animals were vertebrates. Conodont elements are believed to be gill- supporting structures or part of a filter-feeding apparatus. |
Early Jawed Vertebrates
All jawed vertebrates, whether extinct or living, are collectively called gnathostomes (“jaw mouth”) in contrast to the jawless vertebrates, the agnathans (“without jaw”). Living agnathans, the naked hagfishes and lampreys, also are often called cyclostomes (“circle mouth”). The gnathostomes are a monophyletic group since presence of jaws is a derived character state shared by all jawed fishes and tetrapods. Agnathans, however, are defined principally by the absence of a feature—jaws—that characterize the gnathostomes. Therefore the superclass Agnatha may be paraphyletic.
The origin of jaws was one of the most important events in vertebrate evolution. The utility of jaws is obvious: they allow predation on large and active forms of food not available to jawless vertebrates. Ample evidence suggests that jaws arose through modifications of the first two of the serially repeated cartilaginous gill arches. We can see the beginnings of this trend in some ostracoderms where the mouth becomes bordered by strong dermal plates that could be manipulated somewhat like jaws with the gill arch musculature. Later, the anterior gill arches became hinged and bent forward into the characteristic position of vertebrate jaws. Evidence for this remarkable transformation is threefold. First, both gill arches and jaws form from upper and lower bars that bend forward and are hinged in the middle (Figure 25-16). Second, both gill arches and jaws are derived from neural crest cells rather than from mesodermal tissue, the source of most bones. Third, the jaw musculature is homologous to the original gill support musculature. Nearly as remarkable as this drastic morphological remodeling is the subsequent evolutionary fate of jawbone elements—their transformation into ear ossicles of the mammalian middle ear.
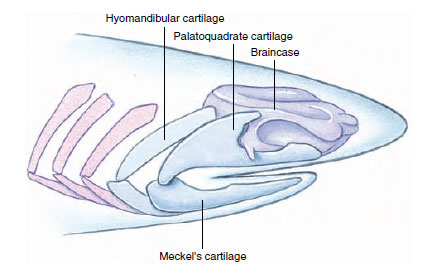 |
| Figure 25-16 How vertebrates got their jaw. The resemblance between jaws and the gill supports of the primitive fishes such as this carboniferous shark suggests that the upper jaw (palatoquadrate) and lower jaw (Meckel’s cartilage) evolved from structures that originally functioned as gill supports. The gill supports immediately behind the jaws are hinged like jaws and served to link the jaws to the braincase. Relics of this transformation are seen during the development of modern sharks. |
Among the first jawed vertebrates were the heavily armored placoderms (plak´cute;avo-derm) (Gr. plax, plate, + derma, skin). These first appear in the fossil record in the early Devonian period (Figure 25-17). Placoderms evolved a great variety of forms, some very large (one was 10 m in length!) and grotesque in appearance. They were armored fish covered with diamondshaped scales or with large plates of bone. All became extinct by the end of the Paleozoic era and appear to have left no descendants. However, the acanthodians (Figure 25-17), a group of early jawed fishes that were contemporary with the placoderms, may have given rise to the great radiation of bony fishes that dominate the waters of the world today.
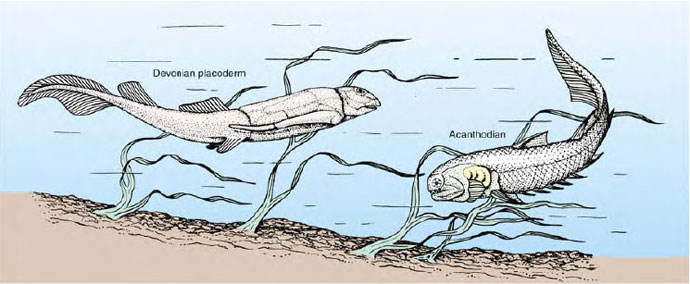 |
| Figure 25-17 Early jawed fishes of the Devonian period, 400 million years ago. Shown are a placoderm (left) and a related acanthodian (right). Jaws and the gill supports from which the jaws evolved develop from neural crest cells, a diagnostic character of vertebrates. Most placoderms were bottom dwellers that fed on detritus although some were active predators. Acanthodians, the earliest-known true jawed fishes, carried less armor than placoderms. Most were marinebut several species entered fresh water. |
Evolution of Modern Fishes and Tetrapods
Reconstruction of the origins of the vast and varied assemblage of modern living vertebrates is based, as we have seen, largely on fossil evidence. Unfortunately the fossil evidence for the earliest vertebrates is often incomplete and tells us much less than we would like to know about subsequent trends in evolution. Affinities become much easier to establish as the fossil record improves. For instance, the descent of birds and mammals from early tetrapod ancestors has been worked out in a highly convincing manner from the relatively abundant fossil record available. By contrast, the ancestry of modern fishes is shrouded in uncertainty.
Despite the difficulty of clarifying early lines of descent for vertebrates, they are clearly a natural, monophyletic group, distinguished by a large number of shared characteristics. We still do not know, however, from which chordate group the vertebrate lineage originated. Early in their evolution, vertebrates divided into agnathans and gnathostomes. These two groups differ from each other in many fundamental ways, in addition to absence of jaws in the former group and their presence in the latter. The appearance of both jaws and paired fins were major innovations in vertebrate evolution, among the most important reasons for the subsequent major radiations of vertebrates that produced the modern fishes and all of the tetrapods, including you, the reader of this section.
Traditional Linnean
Classification of Living Members of Phylum Chordata
Phylum Chordata
-
Group Protochordata (Acrania) Subphylum Urochordata (u'rokor-
da'ta) (Gr. oura, tail, + L. chorda, cord, + ata, characterized
by) (Tunicata): tunicates. Notochord
and nerve cord in freeswimming
larva only; ascidian
adults sessile, encased in tunic. Subphylum Cephalochordata (sef'a-lo-kor-da'ta) (Gr. kephalƒ,
head, + L. chorda, cord): lancelets (amphioxus). Notochord
and nerve cord found along
entire length of body and persist
throughout life; fishlike in form.
-
Subphylum Vertebrata (ver'tebra'ta) (L. vertebratus, backboned).
Bony or cartilaginous vertebrae
surrounding spinal cord; notochord
in embryonic stages, persisting in
some fishes; also may be divided
into two groups (superclasses)
according to presence of jaws.
-
Superclass Agnatha (ag'natha)
(Gr. a, without, +gnathos, jaw) (Cyclostomata): hagfishes,
lampreys. Without true
jaws or paired appendages.(Probably a paraphyletic
group.)
-
Class Myxini (mik-sin'y)
(Gr. myxa, slime): hagfishes. Terminal mouth with
four pairs of tentacles; buccal
funnel absent; nasal sac with
duct to pharynx; 5 to 15
pairs of pharyngeal pouches;
partially hermaphroditic.
Class Cephalaspidomorphi (sef-a-lass'pe-do-morf'e) (Gr. kephale, head, + aspidos, shield, morphe, form) (Petromyzones): lampreys. Suctorial mouth with horny teeth; nasal sac not connected to mouth; seven pairs of pharyngeal pouches.
Superclass Gnathostomata (na'tho-sto'ma-ta) (Gr. gnathos, jaw, + stoma, mouth): jawed fishes, all tetrapods. With jaws and (usually) paired appendages.
-
Class Chondrichthyes (kon drik
'thee-eez) (Gr. chondros,
cartilage, + ichthys, a fish): sharks, skates, rays, chimaeras.
Streamlined body with
heterocercal tail; cartilaginous
skeleton; five to seven gills with
separate openings, no operculum,
no swim bladder.
Class Osteichthyes (ost'eik 'thee-eez) (Gr. osteon, bone,+ ichthys, a fish): bony fishes. Primitively fusiform body but variously modified; mostly ossified skeleton; single gill opening on each side covered with operculum; usually swim bladder or lung. Class Amphibia (am-fib'e-a) (Gr. amphi, both or double, + bios, life): amphibians. Ectothermic tetrapods; respiration by lungs, gills, or skin; development through larval stage; skin moist, containing mucous glands, and lacking scales.
Class Reptilia (rep-til'e-a) (L. repere, to creep): reptiles. Ectothermic tetrapods possessing lungs; embryo develops within shelled egg; no larval stage; skin dry, lacking mucous glands, and covered by epidermal scales. (A paraphyletic group.)
Class Aves (ay'veez) (L. pl. of avis, bird): B. Endothermic vertebrates with front limbs modified for flight; body covered with feathers; scales on feet.
Class Mammalia (ma-may'leea) (L. mamma, breast): mammals. Endothermic vertebrates possessing mammary glands; body more or less covered with hair; well-developed neocerebrum.




