Introduction to Microscope & Comparison of Size & Shape of Microorganisms
Bacterial Shapes and ArrangementsBacteria are unicellular prokaryotic microorganisms. There are 3 common shapes of bacteria: the coccus, bacillus, and spiral. Bacteria divide by binary fission, a process by which 1 bacterium splits into 2.
Coccus
A coccus-shaped bacterium is usually spherical, although some appear oval, elongated, or flattened on one side. Cocci are approximately 0.5 micrometers (µm) in diameter and may be seen, based on their planes of division and tendency to remain attached after replication, in one of the following arrangements:
- Division in one plane produces either a diplococcus or streptococcus arrangement.
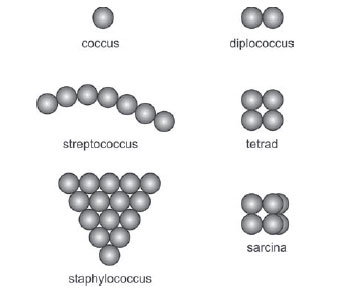
Figure 2 Arrangements of cocci.
(i) Diplococcus: pair of cocci

Figure 1
(ii) Streptococcus: chain of cocci
Figure 1
- Division in 2 planes produces a tetrad arrangement.
A tetrad: square of 4 cocci
- Division in 3 planes produces a sarcina arrangement. Sarcina: cube of 8 cocci.
- Division in random planes produces a staphylococcus arrangement. Staphylococcus: cocci in irregular, often grape-like clusters.
 |
| Figure 5 Tetrad arrangement (appears as a square of 4 cocci shown by arrows). |
As you observe these different cocci, keep in mind that the procedures used in slide preparation may cause some arrangements to break apart or clump together. The correct form, however, should predominate. Also remember that each coccus in an arrangement represents a complete, single, one-celled organism.
Bacillus (rod)
A bacillus or rod is a hotdog-shaped bacterium having one of the following
arrangements:
- bacillus: a single bacillus.
- streptobacillus: bacilli in chains-Streptobacillus arrangement.

Figure 6 Bacilli in chains.
- Coccobacillus: oval and similar to a coccus.
A single bacillus is typically 0.5–1.0 mm wide and 1–4 mm long. Small
bacilli or bacilli that have just divided by binary fission may at first glance be
confused for cocci, so they must be observed carefully. You will, owever, be
able to see bacilli that have not divided and are definitely rod-shaped, as well
as bacilli in the process of dividing.
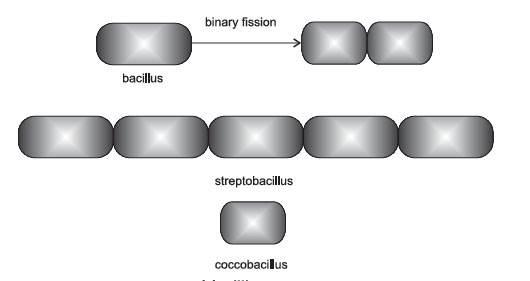
Figure 7 Arrangements of bacilli.
Spiral
Spiral-shaped bacteria occur in one of 3 forms.
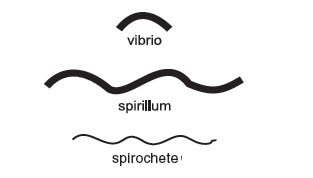 |
| Figure 8 Spiral forms. |
- vibrio: an incomplete spiral, or comma-shaped.
- spirillum: a thick, rigid spiral.
- spirochete: a thin, flexible spiral.
The spirals you will observe range from 5–40 µm long, but some are over
100 µm in length. The spirochetes are the thinnest of the bacteria, often having
a width of only 0.25–0.5 µm.
Yeasts
Yeasts, such as the common baker’s yeast Saccharomyces cerevisiae, are unicellular fungi. They usually appear spherical and have a diameter of 3–5 µm. Yeasts spirochete commonly reproduce asexually by a process called budding. Unlike bacteria, which are prokaryotic, yeast are eukaryotic.
 |
| Figure 9 Saccharomyces cerevisiae (Budding yeast shown by arrows). |
Measurement of Microorganisms
The ocular micrometers provided are calibrated so that when using 1000X oil immersion microscopy, the distance between any 2 lines on the scale represents a length of approximately 1 micrometer. Remember, this does not hold true when using other magnifications.
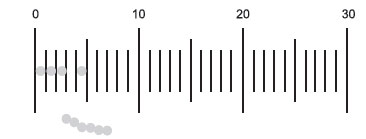 |
| Figure 10 Ocular micrometer. |
The approximate size of a microorganism can be determined using an ocular micrometer, an eyepiece that contains a scale that will appear superimposed upon the focused specimen.
Focusing with Oil Immersion
- Before you plug in the microscope, turn the voltage control dial on the righthand side of the base of the microscope to 1. Now plug in the microscope and turn it on.
- Place the slide in the slide holder, center the slide using the 2 mechanical stage control knobs under the stage on the righthand side of the microscope, and place a rounded drop of immersion oil on the area to be observed.
- Rotate the white-striped 100X oil immersion objective until it is locked into place. This will produce a total magnification of 1000X.
- Turn the voltage control dial on the righthand side of the base of the microscope to 9 or 10. Make sure the iris diaphragm lever in front under the stage is almost wide open (toward the left side of the stage), and the knob under the stage on the lefthand side of the stage controlling the height of the condenser is turned so the condenser is all the way up.
- Watching the slide and objective lens carefully from the front of the microscope, lower the oil immersion objective into the oil by raising the stage until the lens just touches the slide. Do this by turning the coarse focus (larger knob) away from you until the spring-loaded objective lens just begins to spring upward.
- While looking through the eyepieces, turn the fine focus (smaller knob) toward you at a slow steady speed until the specimen comes into focus. (If the specimen does not come into focus within a few complete turns of the fine focus control and the lens is starting to come out of the oil, you missed the specimen when it went through focus. Simply reverse direction and start turning the fine focus away from you.)
- Using the iris diaphragm lever, adjust the light to obtain optimum contrast.
- When finished, wipe the oil off the oil immersion objective with lens paper, turn the voltage control dial back to 1, turn off the microscope, unplug the power cord, and wrap the cord around the base of the microscope.An alternate focusing technique is to first focus on the slide with the yellow-striped 10X objective by using only the coarse focus control and then, without moving the stage, add immersion oil, rotate the white-striped 100X oil immersion objective into place, and adjust the fine focus and light as needed. This procedure is discussed in the introduction to the lab manual.
Specimens
- Prepare slides of the following bacteria:
- Staphylococcus aureus
- Escherichia coli
- Borrelia recurrentis or Borrelia burgdorferi
- Spirillum species.
- Demonstration slides of the following bacteria:
- Micrococcus luteus
- Neisseria gonorrhea
- Streptococcus species
- Bacillus megaterium
- Broth culture of Saccharomyces cerevisiae
- Human hair.
Procedure
- Using oil immersion microscopy (1000X), observe and measure the bacteria
that follow:
Tips for Microscopic Observations
Remember that in the process of making the slide, some of the coccal arrangements will clump together and others will break apart. Move the slide around until you see an area representing the true arrangement of each organism. Also, remember that small bacilli (such as Escherichia coli) that have just divided by binary fission will look similar to cocci. Look carefully for bacilli that are not dividing and are definitely rod-shaped, as well as bacilli in the process of dividing, to confirm the true shape. Also, bacilli do not divide to form clusters. Any such clusters you see are artifacts from preparing the slide. Finally, you will have to look carefully to see the spirochetes, since they are the thinnest of the bacteria. When seen microscopically, spirochetes resemble extremely thin, wavy pencil lines.
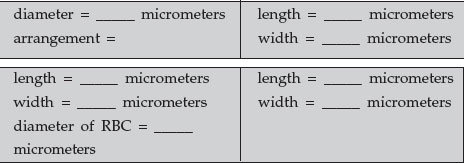
- Staphylococcus aureus: Staphylococcus species, as the genus name implies, have a staphylococcus arrangement (cocci in irregular, often grape-like clusters). Measure the diameter of a single coccus.
- Escherichia coli: Escherichia coli is a small bacillus. Estimate the length and width of a typical rod.
- Borrelia recurrentis: Borrelia species are spirochetes (thin, flexible spirals). You are examining blood infected with Borrelia recurrentis. Measure the length and width of a typical spirochete and the diameter of a red blood cell.
- Spirillum: Spirillum species appear as thick, rigid spirals. Measure the length and width of a typical spirillum. When finished, remove the oil from the prepared slides using a paper towel and return them to their proper tray.
- Observe the demonstration slides of the following bacteria:
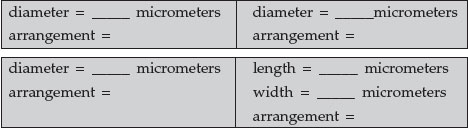
- Micrococcus luteus: Micrococcus luteus can appear as tetrads, cubes of 8, or in irregular clusters. This strain usually exhibits a tetrad or sarcina arrangement. Measure the diameter of a single coccus.
- Neisseria gonorrhea: Neisseria species usually have a diplococcus arrangement. Measure the diameter of a single coccus.
- Streptococcus pyogenes: Streptococcus species, as the genus name implies, usually have a streptococcus arrangement (cocci in chains). Measure the diameter of a single coccus.
- Bacillus megaterium: Bacillus megaterium appears as large bacilli in chains (a streptobacillus). Measure the length and width of a single bacillus.
- Prepare a wet mount of baker’s yeast (Saccharomyces cerevisiae) by putting
a small drop of the yeast culture on a microscope slide and placing a cover
slip over the drop. Using your iris diaphragm lever, reduce the light for
improved contrast by moving the lever almost all the way to the right and
observe using oil immersion microscopy. Measure the diameter of a typical
yeast.
When finished, wash the slide and use it again for step 4. Discard the coverslip in the biowaste disposal container at the front of the room and under the hood. - Remove a small piece of a hair from your head and place it in a small drop of water on a slide. Place a cover slip over the drop and observe using oil immersion microscopy. Measure the diameter of your hair and compare this with the size of each of the bacteria and the yeast observed in steps 1–3. Discard the slide and coverslip in the biowaste disposal containers at the front of the room and under the hood.
- At the completion of the lab, remove the oil from the oil immersion objective, using lens paper, and put your microscope away.
Results
- Make drawings of several of the bacteria from each of the 4 prepared slides and indicate their approximate size in micrometers.
- Make drawings of several of the bacteria from each of the 4 demonstration slides and indicate their approximate size in micrometers.
- Make a drawing of several yeast cells and indicate their size in micrometers.
Saccharomyces cerevisiae
diameter = _____ micrometers. - Make a drawing indicating the size of the bacteria and yeast observed
above, relative to the diameter of your hair.
diameter = _____ micrometers.
 |
 |
Performance Objectives
Discussion
- Name 3 basic shapes of bacteria.
- Name and describe 5 different arrangements of cocci.
- Name and describe 3 different arrangements of bacilli.
- Name and describe 3 different spiral forms.
- Describe the appearance of a typical yeast.
Results
- When given an oil immersion microscope, a prepared slide of a microorganism, and an ocular micrometer, determine the size of that organism in micrometers.
- Using a microscope, identify different bacterial shapes and arrangements.
- Differentiate a yeast from a coccus-shaped bacterium by its size.
- Compare the size of the microorganisms observed in lab with the diameter
of a hair when using oil immersion microscopy.




