Batch Cultures
The most common culture system is the batch culture, due to its simplicity and low cost. This is a closed system, volume-limited, in which there is no input or output of materials, that is, resources are finite. The algal population cell density increases constantly until the exhaustion of some limiting factor, whereas other nutrient components of the culture medium decrease over time. Any products produced by the cells during growth also increase in concentration in the culture medium. Once the resources have been utilized by the cells, the cultures die unless supplied with new medium. In practice this is done by subculturing, that is, transferring a small volume of existing culture to a large volume of fresh culture medium at regular intervals. In this method algal cells are allowed to grow and reproduce in a closed container. A typical batch culture set-up can be a 250 ml Erlenmeyer culture flask with a cotton/gauze bung; in some cases, the bung can be fitted with a Pasteur pipette and air is bubbled into the culture to maintain high levels of oxygen and carbon dioxide and provide mixing.
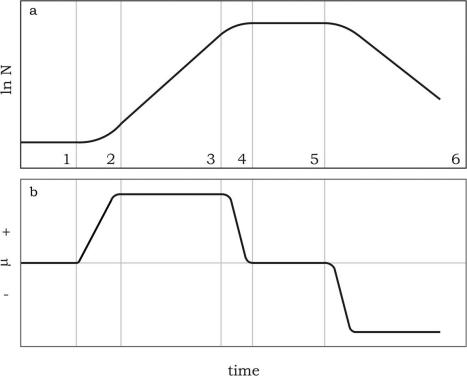
FIGURE 6.1 Growth curve of an algal population under batch culture conditions (a) and corresponding variations of the growth rate (b).
Batch culture systems are highly dynamic. The population shows a typical pattern of growth according to a sigmoid curve (Figure 6.1a), consisting of a succession of six phases, characterized by variations in the growth rate (Figure 6.1b); the six phases are summarized in Table 6.19.

TABLE 6.19 Description of the Six Successive Phases of growth for an Algal Population under Batch Culture Conditions.
The growth curve, relative to the Phases 3, 4 and 5, without the lag, acceleration and crash phases, can be described with a rectangular hyperbolic function similar to the Michaelis-Menten formulation that describes the nutrient uptake kinetics, and the dynamic relationship between photosynthetic rate and irradiance.
After the inoculum, growth does not necessarily start right away, because most cells may be viable, but not in condition to divide. The interval necessary for the transferred cells to adapt to the new situation and start growing is the first phase of the growth curve, the lag phase. This lag or induction phase is relatively long when an algal culture is transferred from a plate to liquid culture. Cultures inoculated with exponentially growing algae have short lag phases, which can seriously reduce the time required for upscaling. The lag in growth is attributed to the physiological adaptation of the cell metabolism to growth, such as the increase of the levels of enzymes and metabolites involved in cell division and carbon fixation. During this phase the growth rate is zero.
After a short phase of growth acceleration, characterized by a continuously increasing growth rate, up to its maximum value, which is achieved in the following exponential phase, the cell density increases as a function of time t according to the exponential function:
where
N2 and
N1 are the number of cells at two successive times and µ is the growth rate. During this phase, the growth rate reached is kept constant. The growth rate is mainly dependent on algal species and cultivation parameters, such as light intensity, temperature, and nutrient availability.
The exponential growth phase normally lasts for a very short period, because cells start to shade each other as their concentration increases. Hence, the culture enters the phase of retardation, and cell growth rate decreases because mainly light, but also nutrients, pH, carbon dioxide, and other physical and chemical factors begin to limit growth. Following this phase, the cell population continues to increase, but the growth rate decreases until it reaches zero, at which point the culture enters the stationary phase, during which the cell concentration remains constant at its maximum value. The final stage of the culture is the death or “crash” phase, characterized by a negative growth rate; during this phase water quality deteriorates, mainly due to catabolite accumulation, and nutrients are depleted to a level incapable to sustain growth. Cell density decreases rapidly
and the culture eventually collapses.
In practice, culture crashes can be caused by a variety of reasons, including the depletion of a nutrient, oxygen deficiency, overheating, pH disturbance, or contamination. The key to the success of algal production is maintaining all cultures in the exponential phase of growth. Also, the nutritional value of the produced algae is inferior once the culture is beyond Phase 4 due to reduced digestibility, deficient composition, and possible production of toxic metabolites.
In batch cultures, cell properties such as size, internal nutrient composition, and metabolic function vary considerably during the above growth phases. This can often make interpretation of the results difficult. During the exponential growth phase, cell properties tend to be constant. However, this phase usually only lasts for a short period of time, and if one wishes to estimate growth rates of the exponential phase of batch cultures, daily sampling appeared to be insufficient to allow a reasonably accurate estimate. Moreover, the accuracy of growth rate determination is highest in artificial, defined media as compared to cells grown in natural surface water media.
A significant advantage of batch culture systems is their operational simplicity. The culture vessels most often consist of an Erlenmeyer flask with a sample to flask volume ratio of about 0.2 in order to prevent carbon dioxide limitation. This volume ratio is only critical if the flasks are shaken by hand once a day during the culturing run. If the flasks are cultured on a rotating shaker table a sample to flask volume ratio of 0.5 is permitted.
Batch culture systems are widely applied because of their simplicity and flexibility, allowing to change species and to remedy defects in the system rapidly. Although often considered as the most reliable method, batch culture is not necessarily the most efficient method. Batch cultures are harvested just prior to the initiation of the stationary phase and must thus always be maintained for a substantial period of time past the maximum specific growth rate. Also, the quality of the harvested cells may be less predictable than that in continuous systems and, for example, vary with the timing of the harvest (time of the day, exact growth phase).
Another disadvantage is the need to prevent contamination during the initial inoculation and early growth period. Because the density of the desired phytoplankton is low and the concentration of nutrients is high, any contaminant with a faster growth rate is capable of outgrowing the culture. Batch cultures also require a lot of labor to harvest, clean, sterilize, refill, and inoculate the
containers.
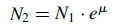 (6.1)
(6.1)





